ENCH 213 Lecture Notes - Confidence Interval, Standard Deviation, Analyte
Document Summary
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
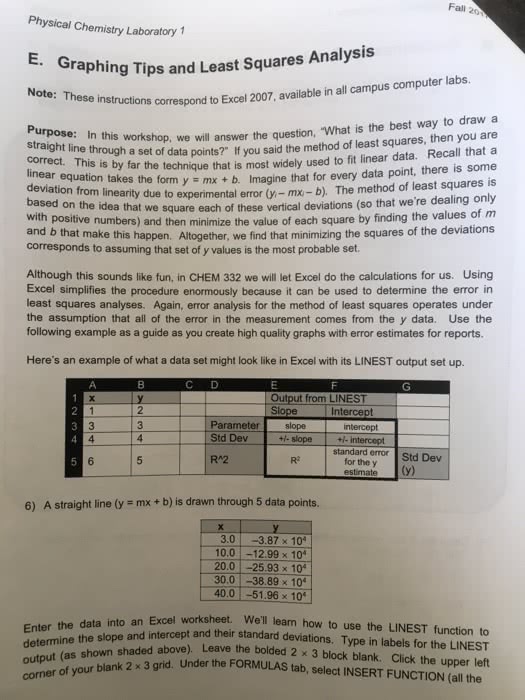
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
Internal Standards
Some analytical methods are subject to one or more common sources of variability in the instrument response. One method aimed at overcoming this variability is the internal standard method. In this method, a known amount of a compound similar to the analyte is added to each standard and sample. Since we know the concentration of the internal standard (IS), we can use the IS response as an indicator of variability. In this type of calibration curve, the quantity plotted on the y-axis is not simply the instrument signal for the analyte, but rather the ratio of the analyte signal to the IS signal.
The IS method is commonly used in gas chromatography, because of the variability inherent in the small volumes injected into the instrument. In the forensic lab, blood alcohol measurements are one of the most common analyses performed. This dry lab example will use blood alcohol data.
You will use ethanol standards that you have purchased. You have 5 of these, with concentrations of 0.050, 0.080, 0.100, 0.200, and 0.300 %w/v of ethanol. You also have a blank, which doesnât contain any ethanol.
You will use n-propanol as the internal standard. You have made up a 0.0300% (v/v) solution of propanol.
You have one sample to run.
You add 2 mL of each of the standards and the sample to GC vials. Then you add 0.450 mL of the internal standard n-propanol to each vial. You get the data below.
| Ethanol concentration, % (w/v) | Peak area for ethanol (pA*sec) | Peak area for n-propanol pA*sec) | Ratio of ethanol area to propanol area | |
First, make a calibration curve using only the area of the ethanol peak and the concentrations of the standards. As in Part 1 a and b, prepare and print a calibration curve including the linear least squares equation of the line equation and the R2 value, and print a copy for your report. Also, calculate the concentration of your sample and the uncertainty (95% confidence interval), as well as the uncertainty of the unknown determination on a percentage basis, and include these values in your report. .
Next, make a calibration curve based on the ratio of ethanol area to propanol area and the concentrations of the standards. As in the previous question, prepare and print a calibration curve including the linear least squares equation of the line equation and the R2 value, and print a copy for your report. Also, calculate the concentration of your sample and the uncertainty (95% confidence interval), as well as the uncertainty of the unknown determination on a percentage basis, and include these values in your report. .
At a blood alcohol concentration of 0.08% (w/v) or more a person is considered impaired. Briefly discuss whether the calculated results for the sample in e and f above support criminal charges of impairment.
Report Requirements:
Spreadsheet with graphs and calculations. Prepare separate sheets in one excel file for each of the following:
Data Set 1
Data Set 2
Calibration with external standard
Standard addition with one standard (optional, could submit hand or type written calculation only)
Standard addition with multiple additions
Internal standard peak areas
Internal standard peak area ratios
Print-outs of graphs
Data Set 1
Data Set 2
Calibration with external standard
Standard addition with multiple additions
Internal standard peak areas
Internal standard peak area ratios
Results
Fill in the table below with results from the spreadsheet. Remember to include units.
| Concentration of unknown | Uncertainty of unknown | Uncertainty of unknown in terms of % | |
| Concentration of unknown | Uncertainty of unknown | Uncertainty of unknown in terms of % | |
| 2d | Concentration of unknown | Uncertainty of unknown | Uncertainty of unknown in terms of % |
| Concentration of unknown | Uncertainty of unknown | Uncertainty of unknown in terms of % | |
| Concentration of unknown | Uncertainty of unknown | Uncertainty of unknown in terms of % | |
Short answers/additional tables
Section 1c:
Section 1d:
Section 1e2:
Section 2a Table:
Section 3b Table:
Section 3b4 calculation
Section 3b5
Section 4d Table with calculated ratios
Section 4g