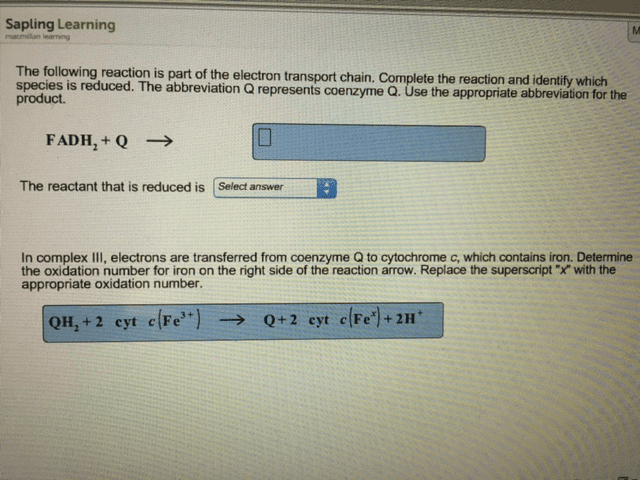
I need help with #8
Acetic acid can be manufactured by reacting methanol with carbon monoxide: CH_3OH (l) + CO(g) rightarrow CH_3COOH (l). (a) Calculate the equilibrium constant for the reaction at 25 degree C. (b) Industrially, this reaction is run at temperatures above 25 degree C. Will an increase in temperature produce an increase or decrease in the mole fraction of acetic acid in an equilibrium mixture? Why are elevated temperatures used for this reaction? Given the following Delta H data for the reactions below, state whether each reaction would be spontaneous at low temperatures, high temperatures, all temperatures, or not at all? Explain. a) PCl_3 (g) + Cl_2 (g) rightarrow PCl_5 (g) Delta H = -88 kJ b) 2 NH_3 (g) rightarrow N_2(g) + 3 H_2(g) Delta H = 92 kJ c) 2 N_2O (g) rightarrow 2 N_2(g) + O_2(g) Delta H = -164 kJ d) H_2O(g) + 1/2 O_2(g) rightarrow H_2O_2 (g) Delta H = 1.6 kJ (a) Write out each nonbalanced reaction below, then above each atom state the oxidation number: and also state which species is being reduced, which species is being oxidized, which species is the reducing agent, and which species is the oxidizing agent. (b) Balance each reaction by the method of half-reactions.