CHEM 126 Lecture Notes - Lecture 9: Molecular Electronic Transition, Equilibrium Constant
Document Summary
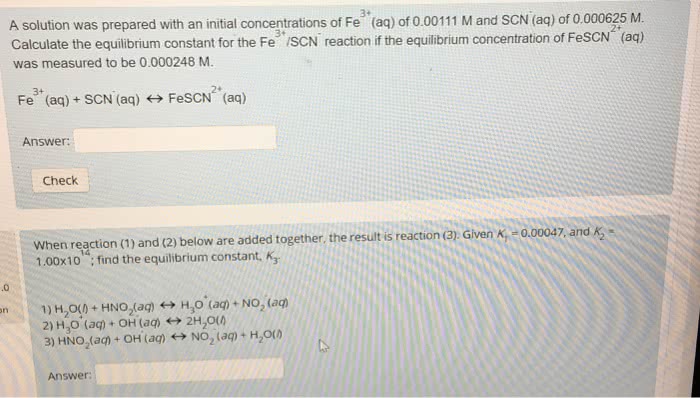
What was the purpose of this experiment going to be, to determine k at various temperatures for the formation of fescn2+ and understand the temperature dependence of keq. Then a linear plot is constructed of ln(k) vs. 1/t to determine ho and so (and go). 5 it was observed that the beer-lambert law was followed to determine the unknown concentration of fescn2+. And so, the concentration of a solution of unknown can be obtained by comparing its absorbance with that of a standard of known. And knowing c2 the equilibrium [fescn2+], and the initial [fe3+] and. Fescn2+ in a standard solution will be almost the same as the initial concentration of scn- in the solution. We do not add a large excess of fe3+ when preparing the solution in part b as the equilibrium will shift to the left to increase the concentration of the reactants: in exp.