CHEM105 Lecture Notes - Lecture 5: Transition State Theory, Reaction Step, Elementary Reaction

15
CHEM105 Full Course Notes
Verified Note
15 documents
Document Summary
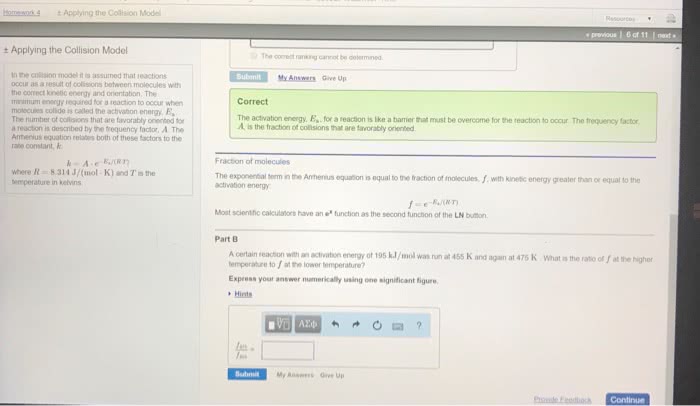
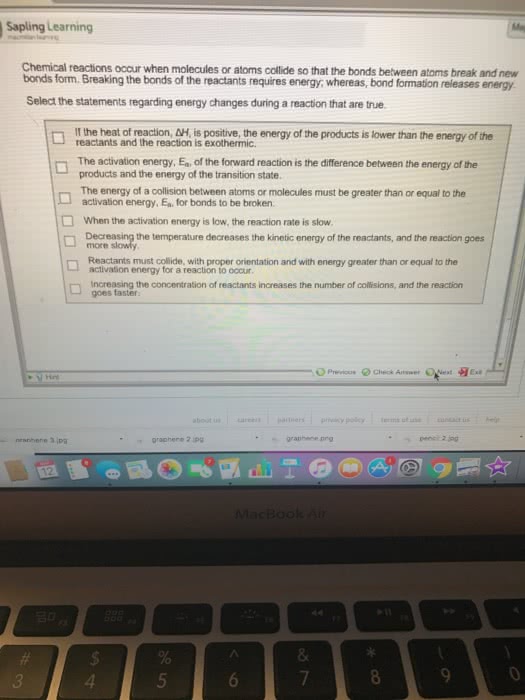
For a reaction with rate = k [a] [b] Rate of reaction = pz x e-ea/rt [a] [b] Rate of reaction = orientation factor x energy factor x collision frequency. This theory depicts the potential energy change of molecules during a collision leading to a reaction. It is assumed that in every reaction (or every step in an overall reaction), a transition state is formed. When molecules approach each other, there is strong repulsion between the electron clouds when they get too close. The molecules must have kinetic energy to overcome this repulsion: the activation energy. An energy diagram is used to show the formation of the transition state. At the highest potential energy, a transition state (t. s. ) (activated complex) is formed. It corresponds to the highest potential energy in a reaction. It is extremely unstable and cannot be isolated.