CHEM261 Lecture : Reactions of Alkanes, Alkenes, and Alkynes

1
CHEM261 Full Course Notes
Verified Note
1 document
Document Summary
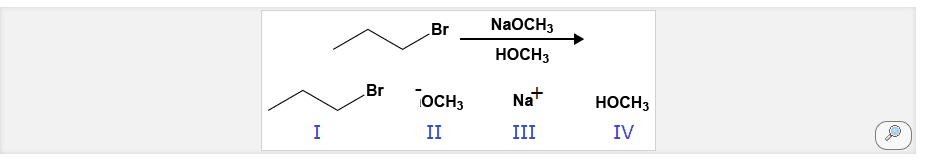
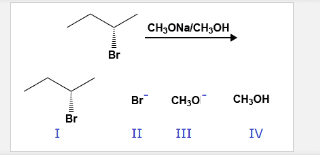
Substrate: must be positive for reaction to happen, electrophile, have positive dipole center, electron sink (low energy empty mo) Sn2 speed is based from [nu] and [s]. There is no reaction intermediate and only 1 ts. The nucleophile attacks the anti-bonding mo of the c attached to the lg. This pushes out the lg, and takes the bond with the c. an inversion of stereochemistry occurs, r to s/s to r if reaction occurs at stereogenic center. If substrate is enantiometically pure, product will remain so (no racemization, remains optically inactive. Sn only works on sp3 c, no vinylic, aromatic, etc. Polar aprotic solvent good for this reaction because is stabilizes ts, faster reaction. Polar protic solvents are capable of h bonding (generally have oh nh bonds) Polar aprotic solvents are polar, but do not h bond (acteonitrile, dmso, hmpa, dimethylformamide) Sn1: the reaction is only dependent on the [s]. It is a 2 step mechanism with 1 intermediate present.