CHEM263 Lecture Notes - Lecture 8: Electrophilic Aromatic Substitution, Benzene, Electrophile
Document Summary
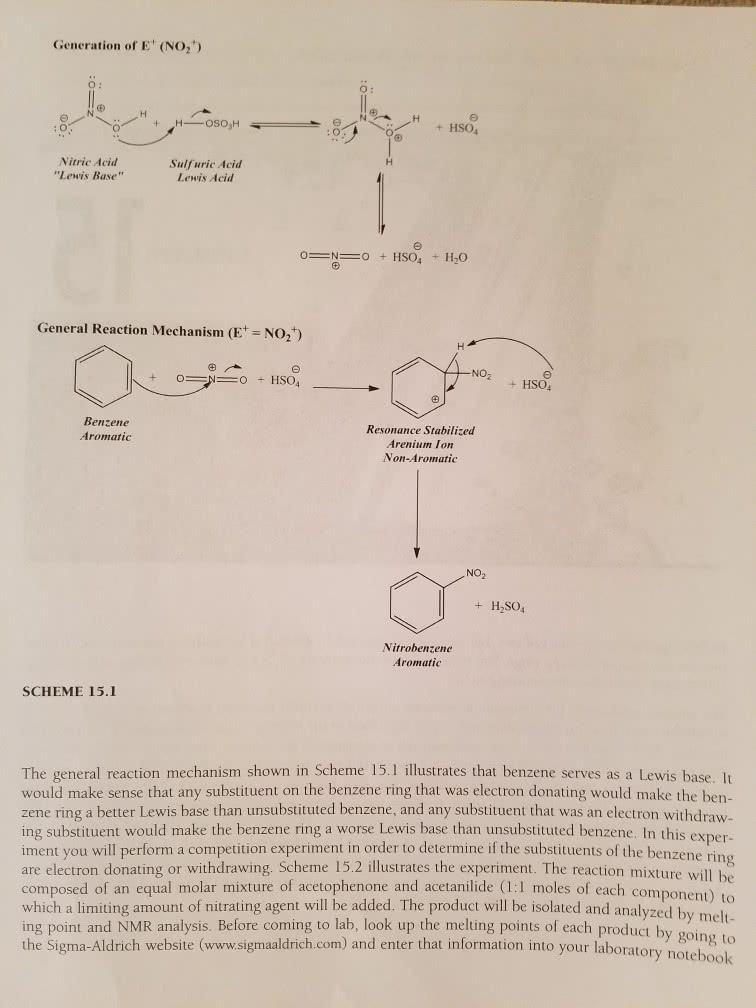
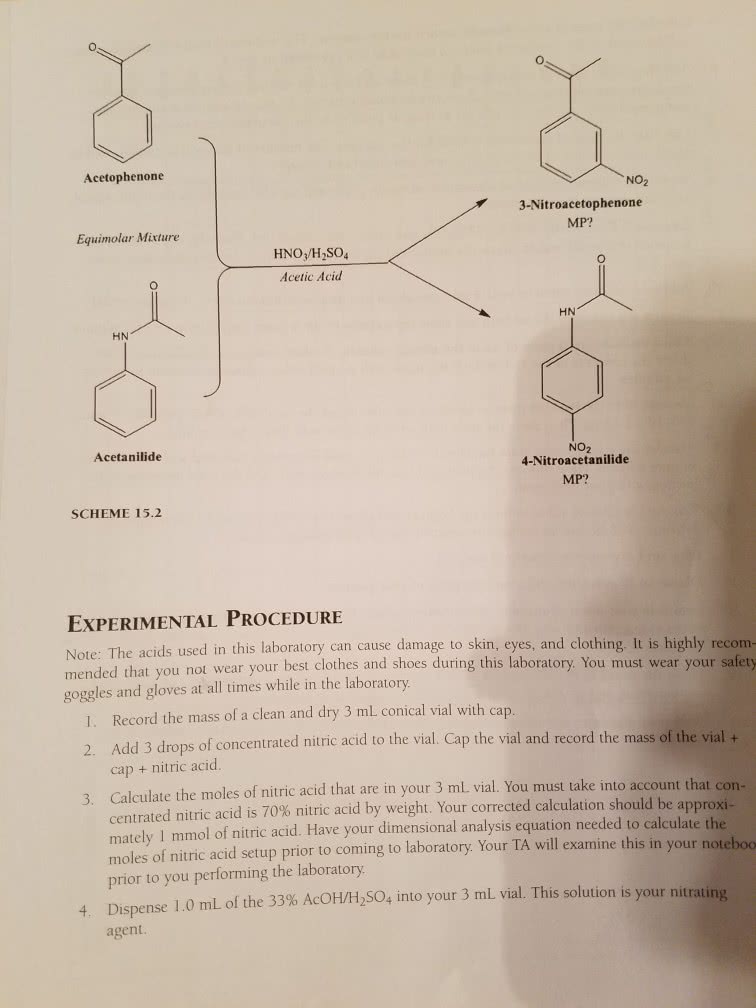
Chem 263 - lecture #8 - electrophilic aromatic substitution for substituted benzenes. Electrophilic aromatic substitution: benzene is very stable (36 kcal/mol) and unreactive compared to alkenes, or even alkanes. Even with light, which will cause bromination in alkanes and alkenes, there is still no reaction for benzene. Lewis acid (electron acceptor) catalyst, benzene can react: some notes on lewis acids. All lewis acids are bronsted acids (proton donor) but not all bronsted. Nucleophile: + centre seeking or donates electrons: mechanism of bromination. Is there an even more general mechanism: yes! In this case, e+ becomes the electrophile, and the system of benzene is the nucleophile. Electrophilic aromatic substitution for substituted benzenes: substituent can donate or withdraw electrons in 2 different ways. Increase donation = increase electron density = increase reactivity. Resonance effects: donate/withdraw electrons through bonds. Electron donating (lone pair of electrons) is o,p. Inductive effects: electronegativity of atom and bond polarity in functional groups.