CHEM 1050 Lecture 4: Guided Reading 4 -Thermo Part4 - Week 4
Document Summary
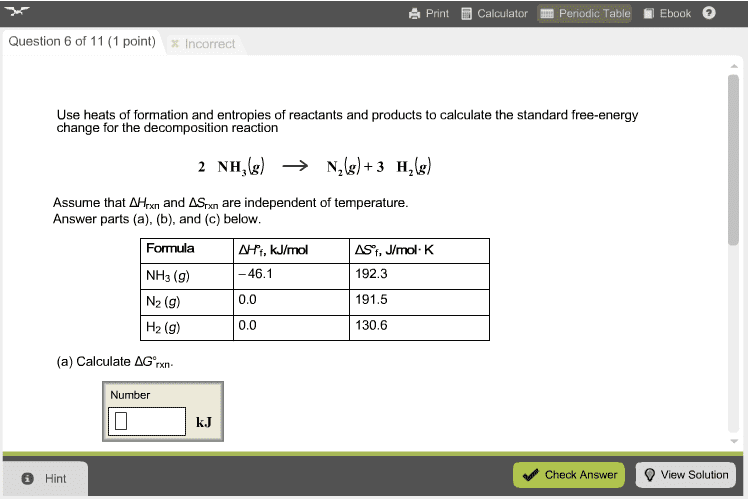
18. 3 standard entropies and the third law of thermodynamics. # state the situations in which the entropy usually increases. # predict the sign of the entropy change of a reaction. # express the standard change of entropy of a reaction in terms of standard entropies of products and reactants. # define the standard free energy of formation, g . # calculate g from standard free energies of formation. # state the rules for using g as a criterion for spontaneity. # relate the free-energy change to maximum useful work. # describe how the free energy changes during a chemical reaction. # write the expression for a thermodynamic equilibrium constant. # indicate how the free-energy change of a reaction and the reaction quotient are related. # relate the standard free-energy change to the thermodynamic equilibrium constant. # calculate k from the standard free-energy change (molecular equation). # calculate k from the standard free-energy change (net ionic equation).