CHEM 1300 Lecture Notes - Lecture 3: Photon, Energy Level, Continuous Spectrum

Lecture 3
Spectra
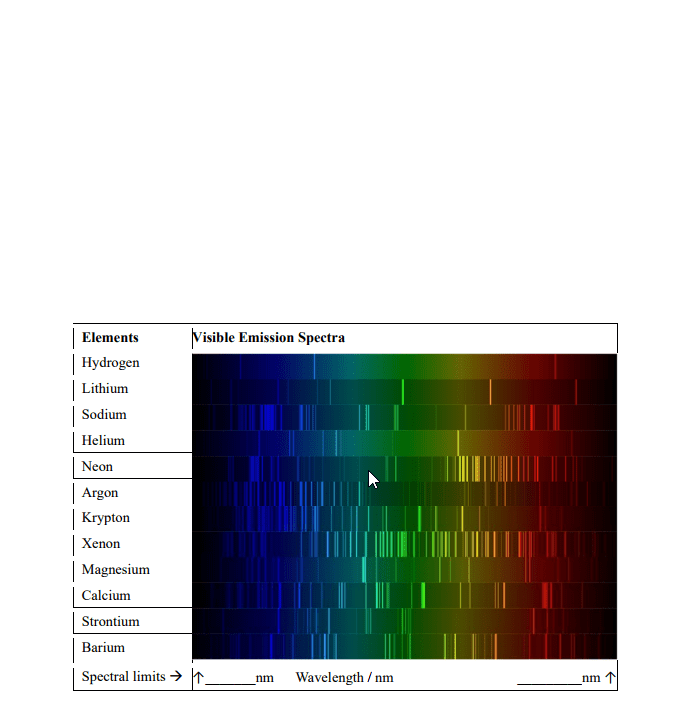
●Dark and light bands
oDiscontinuous spectrum – gaps between the emitted wavelength
▪Absorbed or emitted by an element compound
●Eg: H, He, Ne lights
oContinuous spectrum – absorbs or emits all sized wavelengths
▪No gaps between the wavelength
▪Water droplets
●With abosorption:
●The election may be ejected: is no longer associated with the atom
oProcess called ionization E
▪E- may or may not have kinetic energy
▪Eatom= 0 joules
▪Eionization = Einfinity – E1
▪First energy level: 2.18x10-18J for H
●When the energy is transferred:
oExcited state: any e- NRG level that is not the lowest avail
oGround state: lowest available NRG level
oAs electrons move they spit out wavelengths
●Emission: e- returns to ground state or any lower excited state
●Wavelength will be dependent on the NRG levels of the e-
Theoretical calculation of spectrum EMR
NRG quantization and levels
●The amount of energy that is required for ionization may be related to the atom
oIonization: the process of an atom losing or gaining electrons to become a cation or
anion with a positive or negative charge
oRelated to the atom because of the energy levels or where the e- is potentially located
●With eq’n: positive value means gain energy or absorption, final level is greater than the start
oNegative value means emission and final level is less than when we started
●The amount of energy is specified by the location of the e- where there is (which orbital)
oGround state vs. all other excitatory states
●Rydberg eq’n (see notes)
●The change in energy of an atom is equivalent to the final energy minus the initial energy
●Energy level is equal to -2.18x10-18J/n2 (where n is the NRG level)
o-2.18x10-18J = smallest NRG level possible
●When a wavelength collides with an atom, initial wavelength is incident light
oCan be measured
oe- within orbital will jump up energy levels and emit a wavelength
▪Typically, the NRG of an e- will reflect the NRG of a wavelength
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Dark and light bands: discontinuous spectrum gaps between the emitted wavelength, absorbed or emitted by an element compound. Eg: h, he, ne lights: continuous spectrum absorbs or emits all sized wavelengths, no gaps between the wavelength, water droplets. When the energy is transferred: excited state: any e- nrg level that is not the lowest avail, ground state: lowest available nrg level, as electrons move they spit out wavelengths. Emission: e- returns to ground state or any lower excited state. Wavelength will be dependent on the nrg levels of the e- With eq"n: positive value means gain energy or absorption, final level is greater than the start: negative value means emission and final level is less than when we started. The amount of energy is specified by the location of the e- where there is (which orbital: ground state vs. all other excitatory states. The change in energy of an atom is equivalent to the final energy minus the initial energy.