CHEM 121 Lecture Notes - Lecture 2: Bond Order, Helium Dimer, Electronegativity

79
CHEM 121 Full Course Notes
Verified Note
79 documents
Document Summary
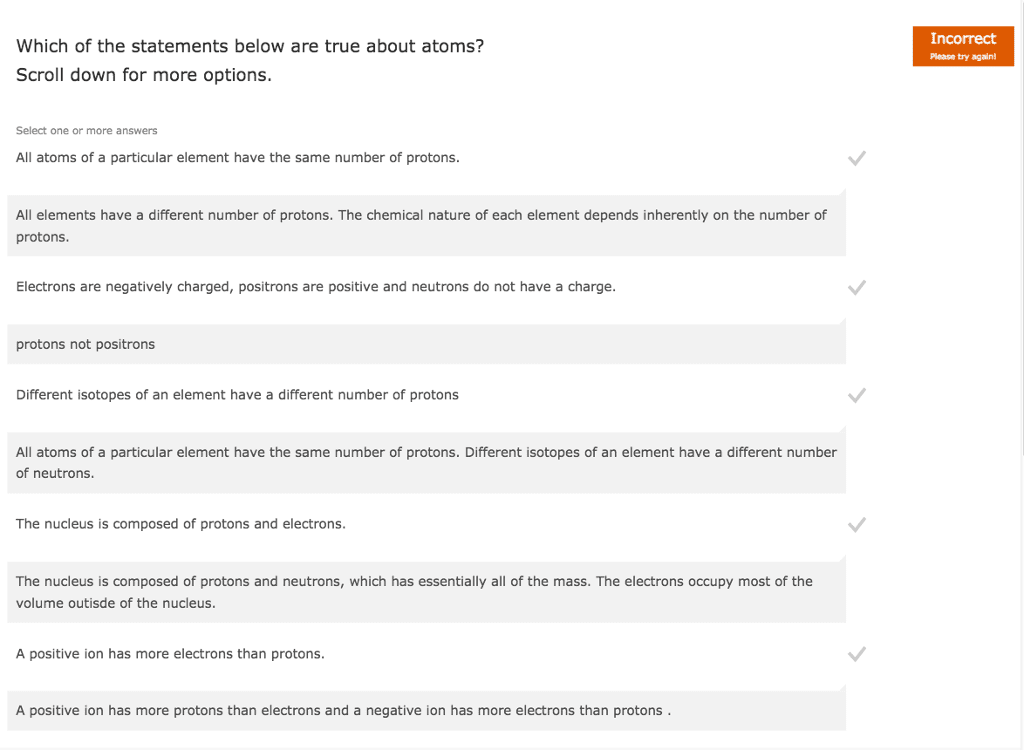
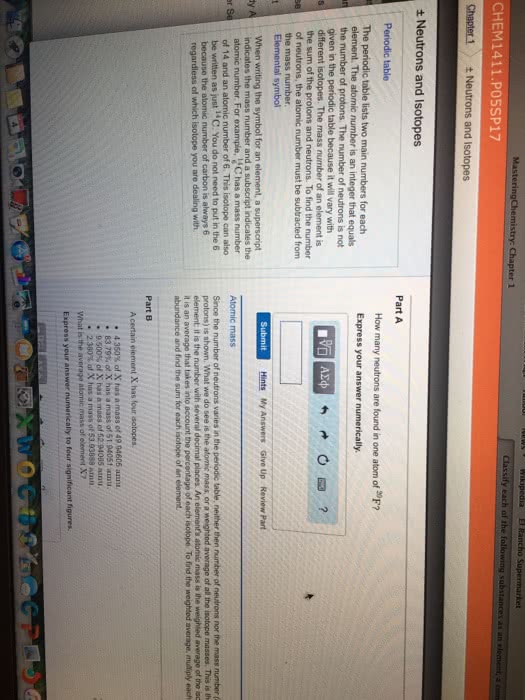
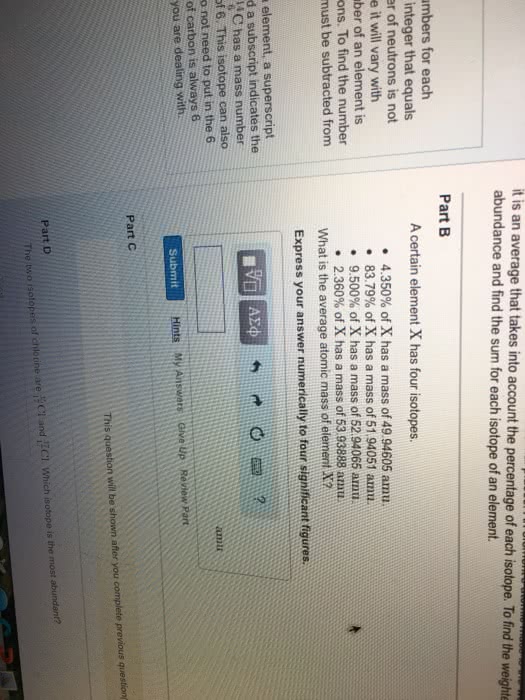
Chem121 - lecture #2 - review of high school. Nucleus contains protons (1+, ~1 a. m. u), neutrons (0 charge, ~1 a. m. v. ) Electrons (1- charge, ~0 a. m. v, more precise: ~0. 00055 a. m. v) All mass is in the nucleus, rest of the space is electrons. All chemistry occurs because of the electrons. Each element has a different atomic number (z) Number of protons+number of electrons=mass number (a) Ie. 12c has 6 neutrons and 6 protons. 13c has 6 protons and 7 neutrons. Proton (atomic number) is always the same for that element. Atomic mass- shown on periodic table is the average for all of the isotopes of that element. S block: first two columns (families) on the left. Interaction between atoms that leads to a rearrangement of electrons to a more stable state. Covalent: sharing of electrons between atoms, ie hydrogen. Ionic: held together by electrostatic force, transfer of electrons, not shared equally, ie nacl.