CHMA10H3 Lecture 4: 4

36
CHMA10H3 Full Course Notes
Verified Note
36 documents
Document Summary
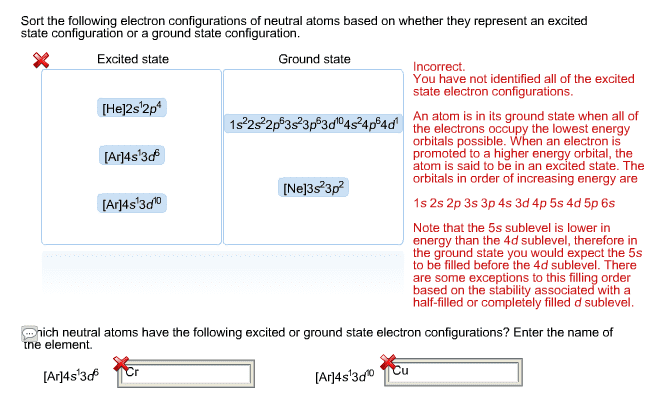
Hund"s rule: energy levels of orbitals: when we are writing electron configuration for atoms, it is important to remember that not all orbitals have the same energy. In fact, orbital energy increases with increasing value of n: when orbitals have equal energy, we call them degenerate. Electron configuration for ions: cationic, formed from removal of electrons, anionic, formed from addition of electrons. Magnetic properties and electron configurations: since electrons are moving electric charges, they will create a magnetic field. If there are unpaired electrons, then a net magnetic field is generated and the species is attracted to an external magnetic field: such species are called paramagnetic. If there are no unpaired electrons, all the magnetic fields cancel out and there is no net magnetic field and the species is slightly repelled by an external magnetic field: such species are called diamagnetic.