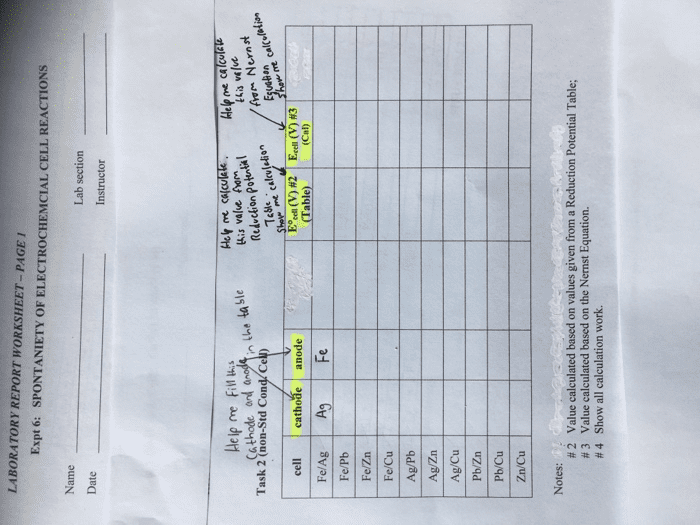
 PLEASE INCLUDE CALCULATIONS
PLEASE INCLUDE CALCULATIONS
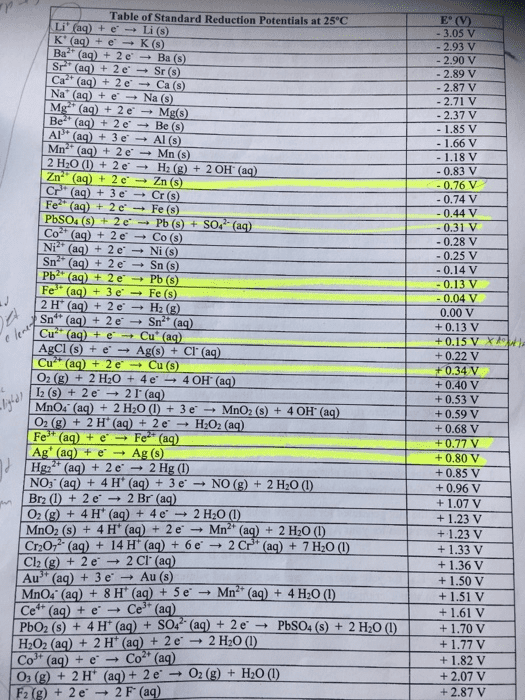
3. Suppose you have the following reagents available under standard conditions (i.e., P- 1 atm for all gases25 °C: C mol L1 for all aqueous species, including H.)
Co(s), Ag (aq), CI(aq), Cr(s), BrO3 (a), 2(s), F(aq)
Consult the standard reduction potentials on the next page and then answer the following questions
(a) Which is the strongest oxidizing agent?
(b) Which is the strongest reducing agent?
PLEASE INCLUDE CALCULATIONS/ FORMULAS
(c) Which of the available reagent(s), if any, will oxidize Cd(s) without oxidizing Pb(s)? If none of the available reagents can be used for this purpose, then identify another reagent from the table that could be used. For one of the reagents you've identified, write down a balanced chemical equation for the reaction that occurs.
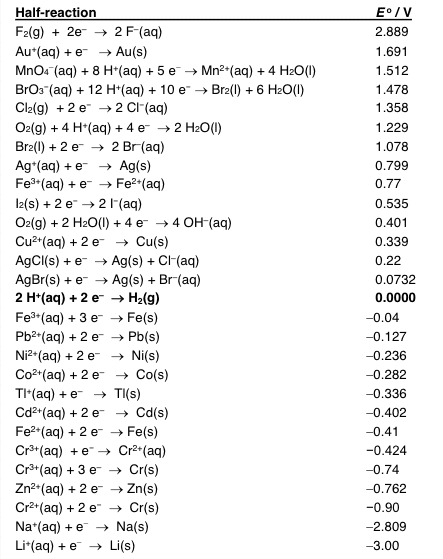
Half-reaction F2(g) + 2e-â 2 F-(aq) Au+(aq) + e--> Au(s) BrOs"(aq) + 12 HTaq) + 10 e--> Br2(l) + 6 H2O(l) Cl2(g) 2 e -2 CI-(aq) Oz(g) + 4 H+(aq) + 4 e-â 2 H2O(l) Br2)2 e2 Br(aq) Ag+(aq) + e-â Ag(s) Fe3+(aq) + e-â Fe2+(aq) 12(s) + 2 e-â 2 I-(aq) 02(g) + 2 H2O(l) + 4 e-â 4 OH-(aq) Cu2+(a)2eCu(s) AgCI(s) eAg(s)Cl-(aq) AgBr(s) + e-â Ag(s) + Braq) 2 H+(aq) + 2 e-â H2(g) Fe (aq)3 e Fe(s) Pb2+(aq) + 2 e-â Pb(s) Ni2*(aq) +2 eNi(s) Co2+(aq) + 2 e--> Co(s) TI"(aq) + e-âTI(s) Cd2+(a)2eCd(s) Fe2(aq)2eFe(s) 2.889 1.691 1.512 1.478 1.358 1.229 1.078 0.799 0.77 0.535 0.401 0.339 0.22 0.0732 0.0000 -0.04 -0.127 -0.236 -0.282 -0.336 -0.402 -0.41 Cr3+(aq) 3e - Cr(s) Zn2 (aq) 2 e -Zn(s) Cr2*(aq) +2 eCr(s) Na*(aq) Na(s) Li (aq) eLi(s) -0.424 -0.74 -0.762 -0.90 -2.809 -3.00
Show transcribed image text Half-reaction F2(g) + 2e-â 2 F-(aq) Au+(aq) + e--> Au(s) BrOs"(aq) + 12 HTaq) + 10 e--> Br2(l) + 6 H2O(l) Cl2(g) 2 e -2 CI-(aq) Oz(g) + 4 H+(aq) + 4 e-â 2 H2O(l) Br2)2 e2 Br(aq) Ag+(aq) + e-â Ag(s) Fe3+(aq) + e-â Fe2+(aq) 12(s) + 2 e-â 2 I-(aq) 02(g) + 2 H2O(l) + 4 e-â 4 OH-(aq) Cu2+(a)2eCu(s) AgCI(s) eAg(s)Cl-(aq) AgBr(s) + e-â Ag(s) + Braq) 2 H+(aq) + 2 e-â H2(g) Fe (aq)3 e Fe(s) Pb2+(aq) + 2 e-â Pb(s) Ni2*(aq) +2 eNi(s) Co2+(aq) + 2 e--> Co(s) TI"(aq) + e-âTI(s) Cd2+(a)2eCd(s) Fe2(aq)2eFe(s) 2.889 1.691 1.512 1.478 1.358 1.229 1.078 0.799 0.77 0.535 0.401 0.339 0.22 0.0732 0.0000 -0.04 -0.127 -0.236 -0.282 -0.336 -0.402 -0.41 Cr3+(aq) 3e - Cr(s) Zn2 (aq) 2 e -Zn(s) Cr2*(aq) +2 eCr(s) Na*(aq) Na(s) Li (aq) eLi(s) -0.424 -0.74 -0.762 -0.90 -2.809 -3.00