CHM135H1 Lecture : D30137.docx
22 views1 pages
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related Documents
Related Questions
The formaldehyde molecule is shown below, and we will assumethat it is in the xy plane.
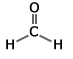
Which orbitals on carbon are involved in the sp2 hybrid orbitalsand which orbital is involved in ??
| 2s, 2pzinvolved in hybridization and2pxinvolved in ? bonding. | |
| The 2s, 2px, 2pyare involvedin hybridization and 2pzis involved in ? bonding. | |
| 2s, 2pxinvolved in hybridization and2pyinvolved in ? bonding. | |
| 2s, 2px, 2pzinvolved inhybridization and 2pyinvolved in ? bonding. |

