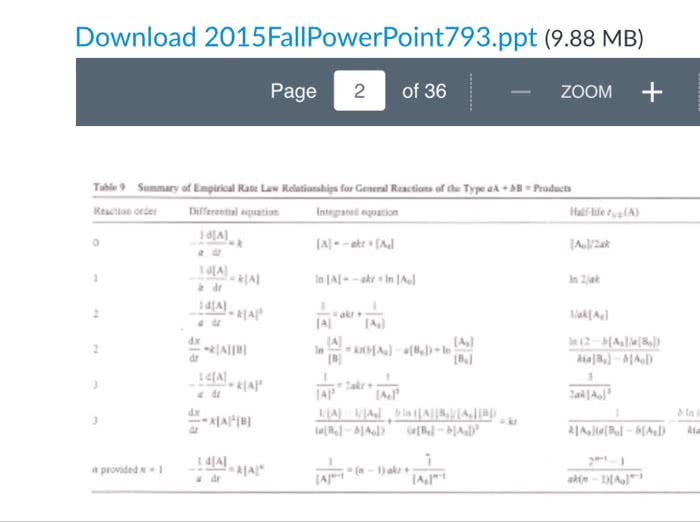
Consider a reaction with the stoichiometry 2A + 3B rightarrow C + 4D. The rate law is first order overall and first order with respect to reactant A. Write the differential form of the rate law with variable [A] and then with conversion variable x. Derive the integrated form of this rate law showing easy-to-follow steps in the derivation. Show that your result matches the result in Table 9 on the Power Point slide. Derive the integrated form of the following rate equation using the Shillady equation. dx/dt = k[A][B] Show easy-to-follow steps in your derivation. Get your integrated equation in a form that matches the Table 9 equation on the Power Point slide. In class, the integrated equation for a 3^rd order, type II equation with stoichiometry 2A + B rightarrow products was derived in detail. However, the last few steps were not all given. Start with the following equality and complete the derivation of the integrated equation showing easy-to-follow steps. Get your integrated equation in a form that matches the Table 9 equation on the Power Point slide.