CHM135H1 Lecture Notes - Lecture 4: Ph Indicator, Inert Gas, Molar Concentration

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
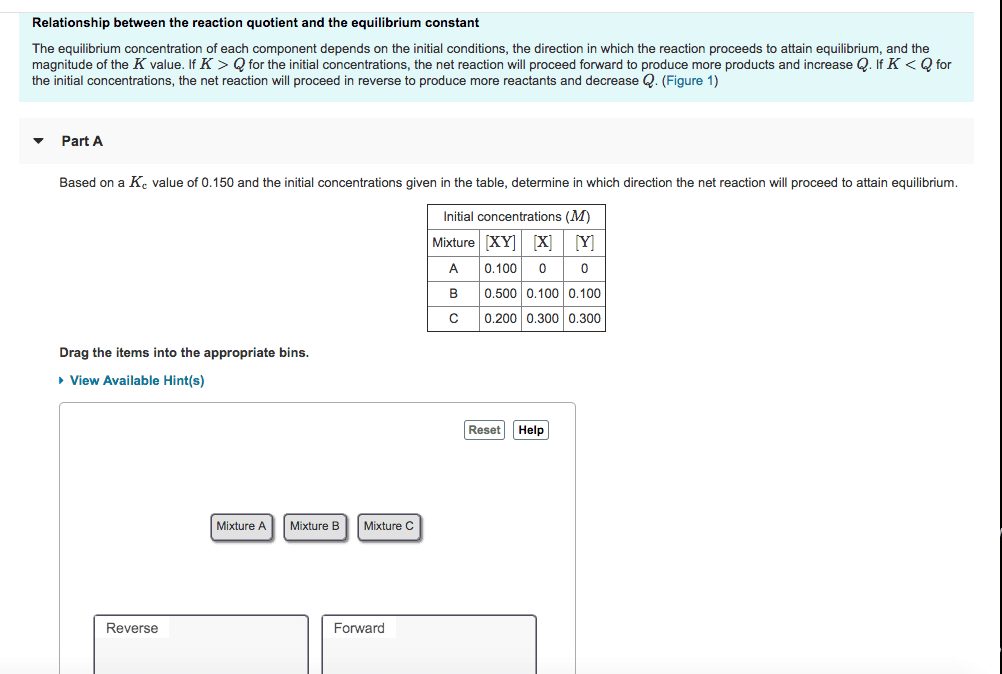
The equilibrium constant is always the same for a given reaction at a given temperature. It is unitless, since each: general definition of equilibrium constant. General reaction equation: (cid:1853)(cid:1827)+(cid:1854)(cid:1828)=(cid:1855)(cid:1829)+(cid:1856)(cid:1830) reverse rate=(cid:1863)(cid:2869)(cid:1863) (cid:2869)=concentration of products forward rate concentration of reagents=equilibrium constant (cid:1837)(cid:3030)=[(cid:1829)](cid:3030)[(cid:1830)](cid:3031) (cid:1866)(cid:3028)(cid:3046) is the number of moles of the products the number of moles of the reagents (determined by (cid:1837)=(cid:1837)(cid:3030)(cid:4666)(cid:4667) (cid:3041), (cid:1866)(cid:3028)(cid:3046)=(cid:1855)+(cid:1856) (cid:1853) (cid:1854) concentration is divided by 1m to cancel out the units: equilibrium involving gases. Kp is unitless, since each concentration is divided by 1 atm to cancel out the units. stoichiometry). All the partial pressures must be in units of atmosphere (not mmhg: equilibrium involving pure solids and liquids. The concentrations of pure solids and liquids are approximately constant (standard concentrations), so they can be omitted. If a stress is applied to a reaction mixture at equilibrium, net reaction occurs in the direction that relieves the stress.