CHM135H1 Lecture : Notes taken during lecture

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
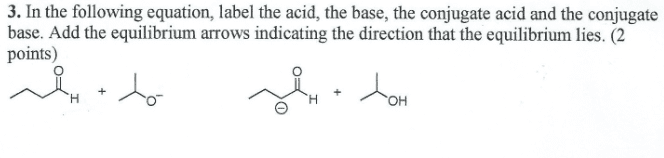
1 k small not many ions all types of equilibrium equations, all have similar behaviour starting at a point not at equilibrium, then tending to equilibrium. Conjugate base is the hs, conjugate acid is the nh4. Conjugate acid-base pairs: the h2s and hs, and the nh3 and nh4. Each acid-base pair related by one proton. Water can both give proton, and receive proton. Acid weaker than h3o+ therefore equilibrium shifts to products" side. Strong acid wants to give protons, strong bases want to receive protons. The stronger the acid, the weaker the conjugate base. Direction of reaction is towards the products" side. Cannot have stronger a or b and weaker a or b on one side. Must be stronger acid and stronger base reacting to form weaker acid and weaker base the ion-product constant for water holds for any aqueous solution if either of [h3o+] or [oh-] increases, the other decreases. Water acts as base to accept proton.