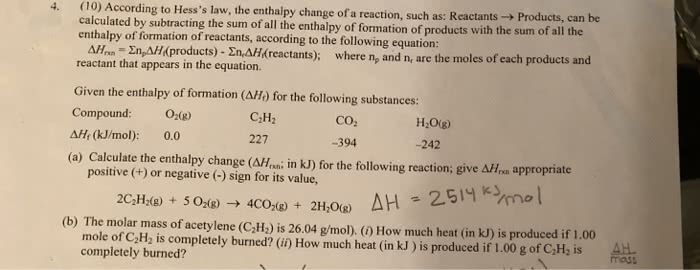Chemistry 1302A/B Lecture Notes - Lecture 7: Bromine, Exothermic Process
Document Summary
De ned as: the enthalpy change in a reaction in which one mole of a substance is formed from its elements in their standard state (state of substances at 1 atm & 25 c) Hf0 of an element in its standard state is 0. C2h5oh 2 c(graphite,s) + 3 h2(g) + 1/2o2(g) c2h5oh (l) Nh4br 1/2 n2(g) + 2 h2(g) + 1/2 br2(l) Hess"s law states that the enthalpy change of the reaction is equal to the sum of the enthalpy changes for the individual steps of the reaction. Hf0 can be calculated for any reaction that is a combination of other reactions for which. Is the same as the enthalpy change for: Hf0 = - hf0 (reactants) + hf0 (products) or. Hf0 (products) - hf0 (reactants) if h < 0 exothermic if h > 0 endothermic. For a generic reaction: aa + bb cc + dd.