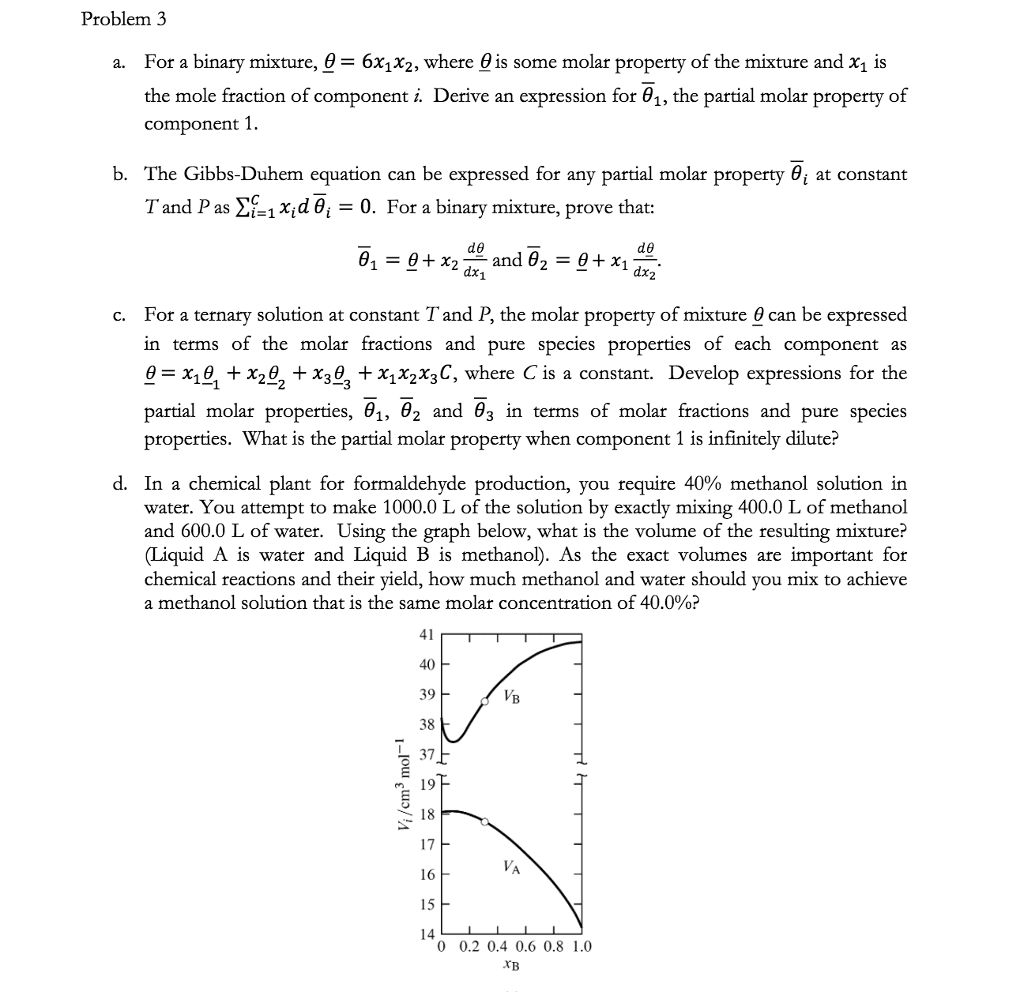
Chemistry 2214A/B Lecture Notes - Lecture 19: Mole Fraction, Partial Molar Property, Gibbs Free Energy
Document Summary
Partial molar gibbs energy (chemical potential: a, b) Partial molar gibbs energy: the gibbs durham equation nad a + nbd a = 0. In general we can write nid i = 0. The implication of this result is that the chemical potential of a component does not change independently; that is that if a increases, b must decrease. Ideal liquid mixture: interaction between a-a is the same as a-b and b-b. It is the mixture of two completely miscible, volatile liquid. The total pressure above the mixture: p = pa + pb= xa pa* + (1 - xa) pb* Liquid vapor phase diagram of a binary liquid mixture at constant temperature. It is a plot of p vs. composition (xa, ya, xa. The total vapor pressure above a mixture of two volatile liquid varies depending on the composition of the mixture (xa and xb). For each total pressure, there are two compositions xa (liquid) and ya(vapor) for each component.