Chemistry 2374A Lecture Notes - Lecture 9: Ideal Gas Law, Thermodynamic Equilibrium, Inexact Differential
Document Summary
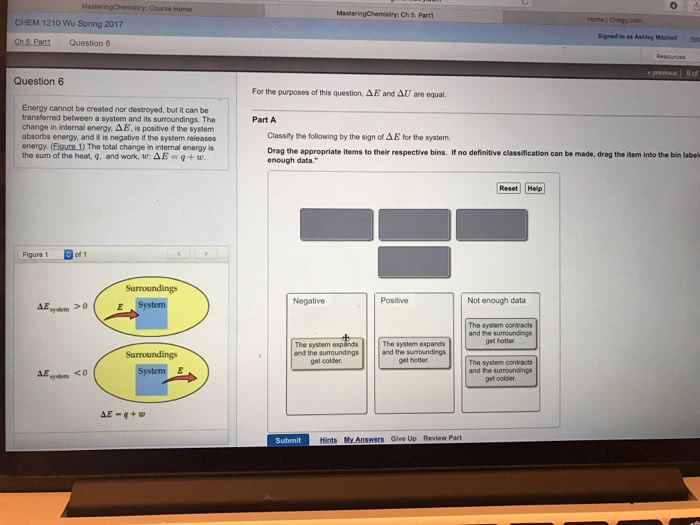
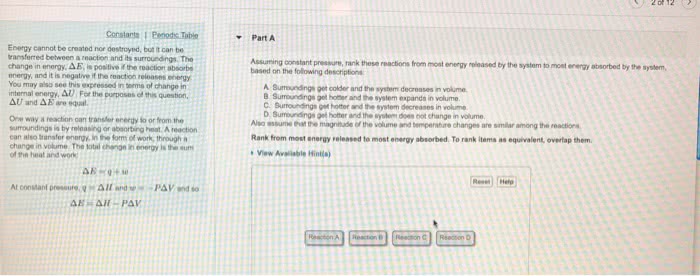
Chapter 2. 1 (cid:498)mathematical(cid:499) version of the first law. The internal energy of u of a system can be changed by heat (q) or work (w) The (cid:498)u(cid:499) is always in the system perspective; the system"s internal energy. The is for big change; d is for a small change. The internal energy u of a system can be changed by heat (cid:498)q(cid:499) or work (cid:498)w(cid:499) The ensuing combustion is highly exothermic, which releases heat therefore, internal. If work is positive, the work is going into the system; verse versa. If heat is positive, the heat is going into the system; verse versa energy is a state function (i. e. exact differential), while heat and work are path functions (i. e. inexact differentials) because integration must account for the path taken. For an infinitesimal change (small change) du = dq + dw d is used for an exact differential; d is used for an inexact differential.