CH111 Lecture Notes - Lecture 10: Activated Complex, Reagent, Second Order (Religious)
Document Summary
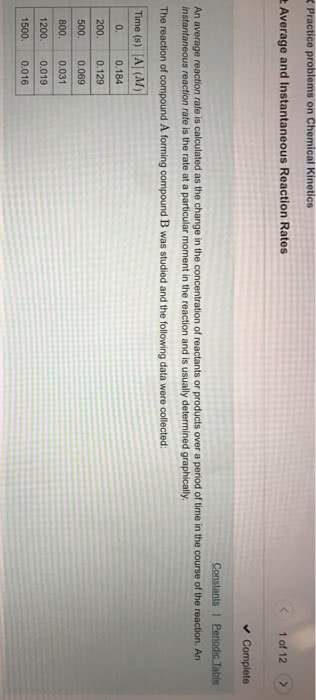
The average rate of reacion is calculated by o. The instantaneous rate is the change in concentraion at any one paricular ime. It is determined by taking the slope of a line tangent to the curve at that paricular point: irst derivaive of the funcion. Nature of the reactants: for solid reactants, the greater the surface area, the faster the rate of reacion. For example: inely divided sawdust will burn faster than a log. Concentraion of the reactants: in soluion, rate of reacion will increase with increasing reactant concentraion, for gases, rate of reacion will increase with increasing pressure. Reacion temperature: reacion rate generally increases considerably with increasing temperature. Presence of a catalyst: catalysts generally increase reacion rate. Rate law (rate equaion): a representaion of the dependence of the rate of reacion on the concentraion of the species: rate = k[a]x[b]y[c]z. Reacion orders can be posiive, negaive, integers or fracions.