CHEM 1000 Lecture Notes - Lecture 6: Azimuthal Quantum Number, Magnetic Quantum Number, Bohr Radius
144 views8 pages
20 Jan 2017
School
Department
Course
Professor
Document Summary
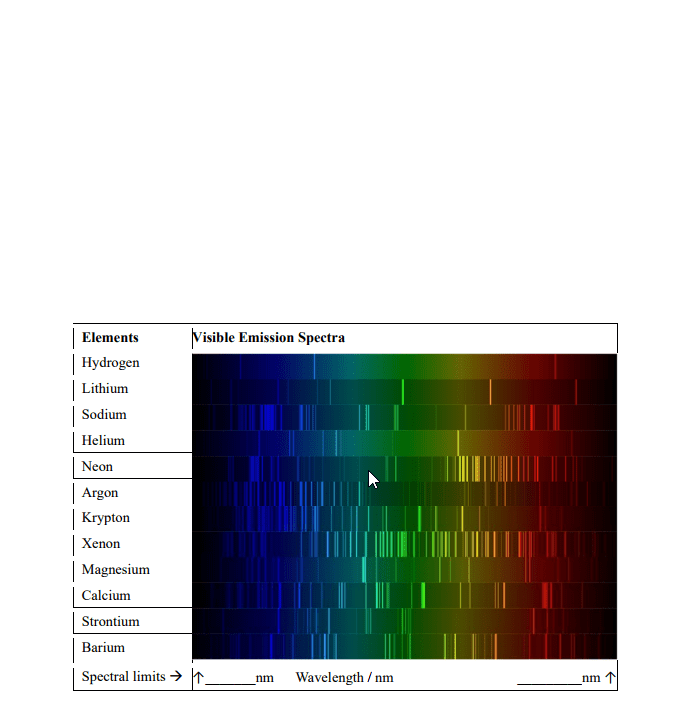
Atomic spectra- the spectra of excited atoms could not be explained by 19th century models of the atom. Spectra were ultimately explained by the quantum theory. Energy has a limited number of discrete values. The energy between two energy values is a quantum of energy. The fundamental principles of the bohr model of the atom are: The electron moves in circular orbits around the nucleus. The electron has constant energy and no energy is emitted. An electron can only go from one allowed orbit to another. Cannot explain emission spectra from multi- electron atoms. Cannot explain the effect of magnetic fields on spectra. No reason for the fixed, quantized orbits. The most important is that of wave- particle duality. The photoelectric effect required that although light was a wave it has particle like properties. De broglie formulated the reverse, the wave particle duality. When the wave length is comparable to the dimensions. When the wavelength is like spacing between atoms.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232