CHEM 142 Lecture Notes - Lecture 6: Chloroform, Ethanol, Sodium Chloride
39 views5 pages
7 Jul 2016
School
Department
Course
Professor
Document Summary
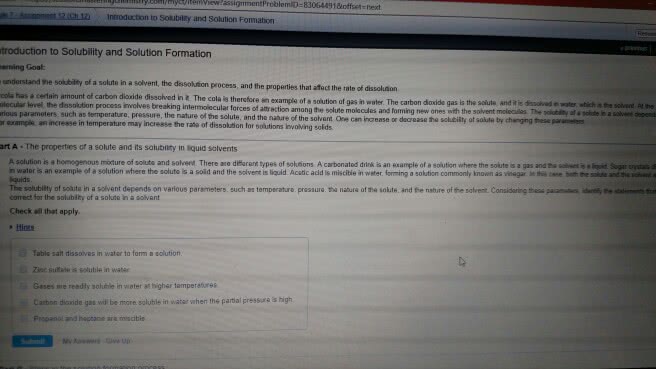
A solution is a homogeneous mixture of at least two components: a solvent and a solute. The solvent is the substance that dissolves and the solute is the substance that is dissolved (literally, is loosened apart ). The solvent is whatever you have more of and determines the state of the solution (so a gas dissolved in water would be a liquid solution think about your soda). Solutions can be any phase (a solid can dissolve in another solid brass and steel are examples). Nature favors processes that maximize disorder consider your bedroom if you do not put in some energy to keep it tidy! For a pure material the only option is to have solvent next to solvent particles or solute next to solute particles. In a solution you can have solute next to solute and solvent next to solve and, but you can also have solute next to solvent.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232