CHEM 142 Lecture Notes - Lecture 9: Reaction Rate Constant, Penicillin, Stoichiometry
88 views7 pages
7 Jul 2016
School
Department
Course
Professor
Document Summary
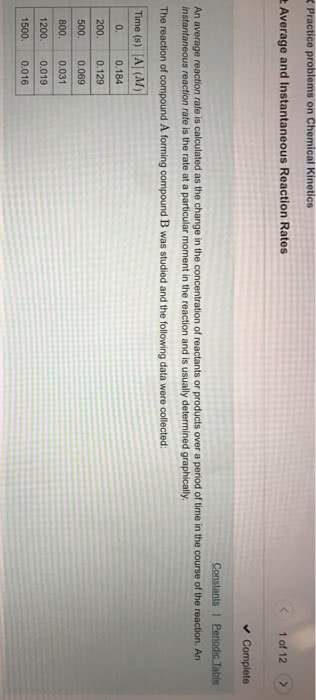
Rate is the change in a measurement over a period of time. In chemistry, we define the rate of a chemical reaction as a change in concentration (in molarity) over time (s). Use the concentration v. time graph for the decomposition of no2 to no and o2 to answer the following questions. 0-50 (0. 008 m-0. 01 m)/ (50s-0s) = -4. 0 10-5 m/s. 150-200 (0. 0048 m-0. 0055 m)/ (200s-150s) = -1. 4 10-5 m/s. 1 originally written by dr. harris, 2016: we can determine the instantaneous rate of change for [no2] at a given point based on the slope of the tangent line at that point. Estimate the rate of change of [no2] when [no2] = 0. 008 m and. 0. 008m (0. 0068-0. 0088)/(80-20) = -3. 3 10-5 m/s. As the concentration of the reactant no2 decreases the rate of the reaction decreases: use the stoichiometry of the reaction to determine the average rate of change for [o2] and [no] from.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232