To understand how to use integrated rate laws to solve for concentration.
A car starts at mile marker 145 on a highway and drives at 55 mi/hr in the direction of decreasing marker numbers. What mile marker will the car reach after 2 hours?
This problem can easily be solved by calculating how far the car travels and subtracting that distance from the starting marker of 145.
55 mi/hrÃ2 hr=110 miles traveled
milemarker 145â110 miles=milemarker 35
If we were to write a formula for this calculation, we might express it as follows:
milemarker=milemarker0â(speedÃtime)
where milemarker is the current milemarker and milemarker0 is the initial milemarker.
Similarly, the integrated rate law for a zero-order reaction is expressed as follows:
[A]=[A]0ârateÃtime
or
[A]=[A]0âkt
since
rate=k[A]0=k
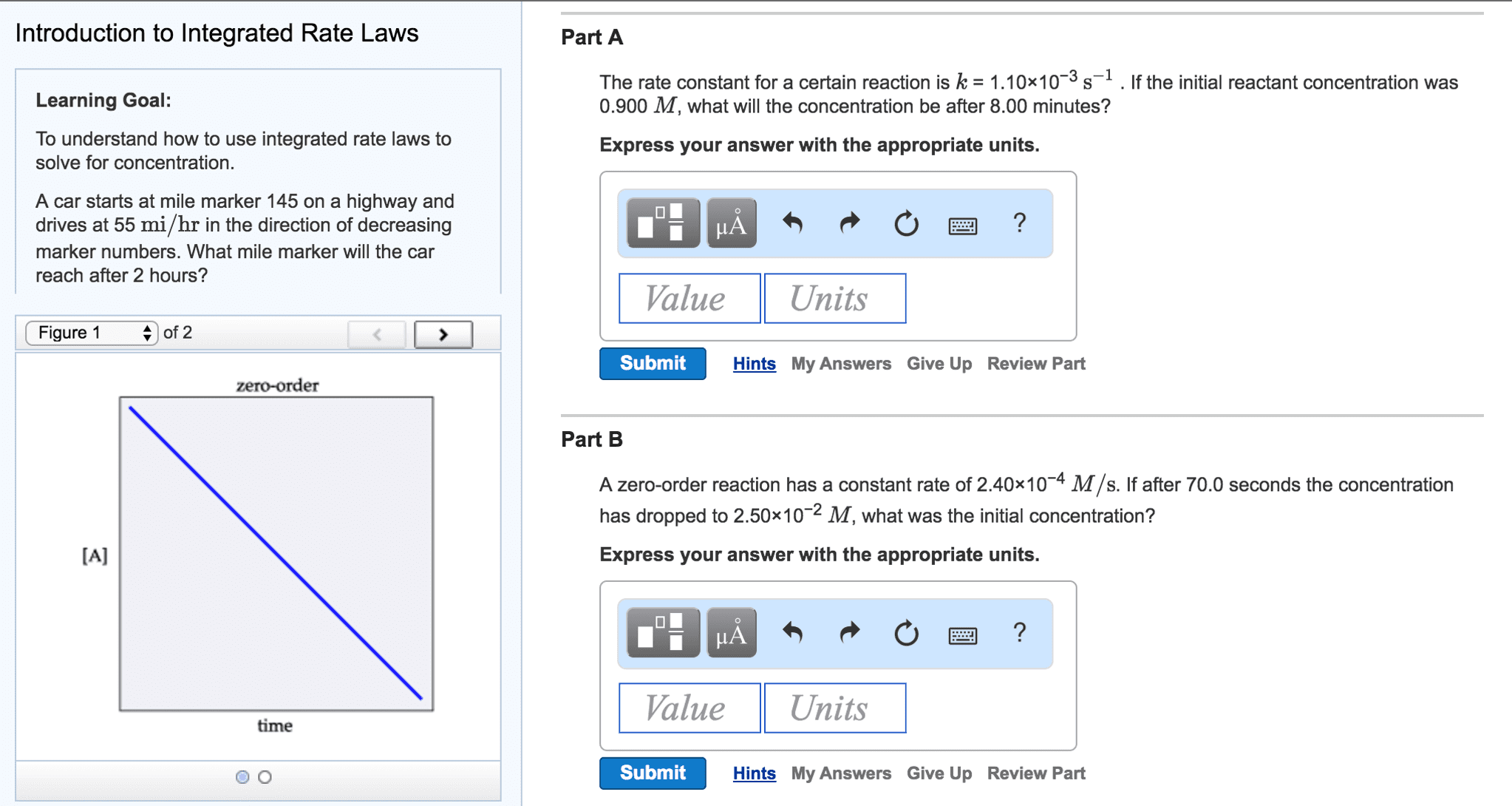
A zero-order reaction (Figure 1) proceeds uniformly over time. In other words, the rate does not change as the reactant concentration changes. In contrast, first-order reaction rates (Figure 2) do change over time as the reactant concentration changes.
Because the rate of a first-order reaction is nonuniform, its integrated rate law is slightly more complicated than that of a zero-order reaction.
The integrated rate law for a first-order reaction is expressed as follows:
[A]=[A]0eâkt
where k is the rate constant for this reaction.
The integrated rate law for a second-order reaction is expressed as follows:
1[A]=kt+1[A0]
where k is the rate constant for this reaction.
PART A
The rate constant for a certain reaction is k = 3.50Ã10â3 sâ1 . If the initial reactant concentration was 0.400 M, what will the concentration be after 5.00 minutes?
Express your answer with the appropriate units.
PART B
A zero-order reaction has a constant rate of 5.00Ã10â4M/s. If after 75.0 seconds the concentration has dropped to 2.50Ã10â2M, what was the initial concentration?
Express your answer with the appropriate units.
To understand how to use integrated rate laws to solve for concentration.
A car starts at mile marker 145 on a highway and drives at 55 mi/hr in the direction of decreasing marker numbers. What mile marker will the car reach after 2 hours?
This problem can easily be solved by calculating how far the car travels and subtracting that distance from the starting marker of 145.
55 mi/hrÃ2 hr=110 miles traveled
milemarker 145â110 miles=milemarker 35
If we were to write a formula for this calculation, we might express it as follows:
milemarker=milemarker0â(speedÃtime)
where milemarker is the current milemarker and milemarker0 is the initial milemarker.
Similarly, the integrated rate law for a zero-order reaction is expressed as follows:
[A]=[A]0ârateÃtime
or
[A]=[A]0âkt
since
rate=k[A]0=k
A zero-order reaction (Figure 1) proceeds uniformly over time. In other words, the rate does not change as the reactant concentration changes. In contrast, first-order reaction rates (Figure 2) do change over time as the reactant concentration changes.
Because the rate of a first-order reaction is nonuniform, its integrated rate law is slightly more complicated than that of a zero-order reaction.
The integrated rate law for a first-order reaction is expressed as follows:
[A]=[A]0eâkt
where k is the rate constant for this reaction.
The integrated rate law for a second-order reaction is expressed as follows:
1[A]=kt+1[A0]
where k is the rate constant for this reaction.
PART A
The rate constant for a certain reaction is k = 3.50Ã10â3 sâ1 . If the initial reactant concentration was 0.400 M, what will the concentration be after 5.00 minutes?
Express your answer with the appropriate units.
PART B
A zero-order reaction has a constant rate of 5.00Ã10â4M/s. If after 75.0 seconds the concentration has dropped to 2.50Ã10â2M, what was the initial concentration?
Express your answer with the appropriate units.