CHM 3411L Lecture Notes - Lecture 3: Chemical Bond, Scalar Multiplication, Covalent Bond
Document Summary
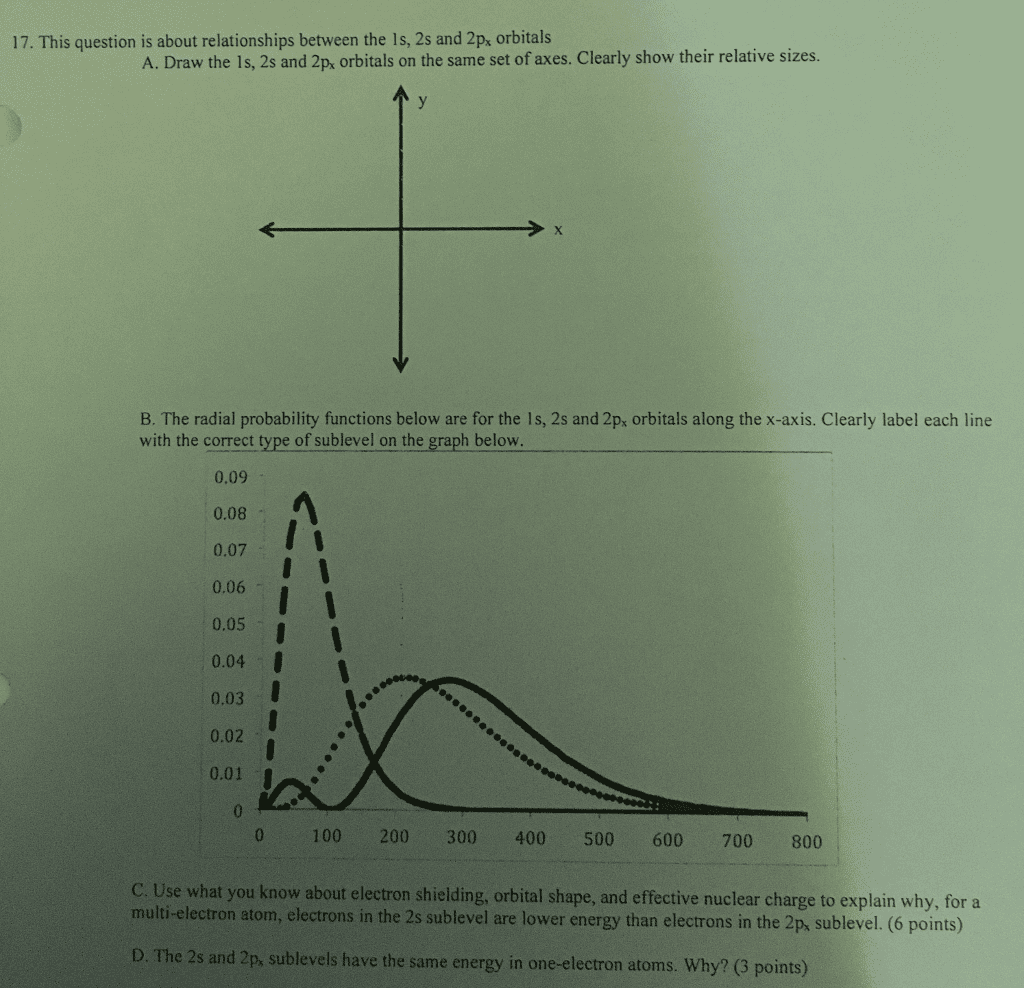
True or false; energy in a 2s orbital is higher than energy of a 1s orbital. What is the orbital state of li and be? (3) li: 1s 2s (4) be: 1s 2s . Pz (popping out of the plane of the page, towards and away from you) True or false; the 3 different orientations do not have the same amount of energy. All 3 orientations have the same amount of energy. The 2p orbital only has one node (one point of 0 electron density) True or false; energy in a 2p orbital is higher than energy of a 2s orbital. The 2p orbital allows carbon to form more than one bond with itself. What is hunds rule? every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied, and all electrons in singly occupied orbitals have the same spin.