CHEM 3650 Lecture Notes - Lecture 3: Endergonic Reaction, Adrenal Cortex, Acid Dissociation Constant
Document Summary
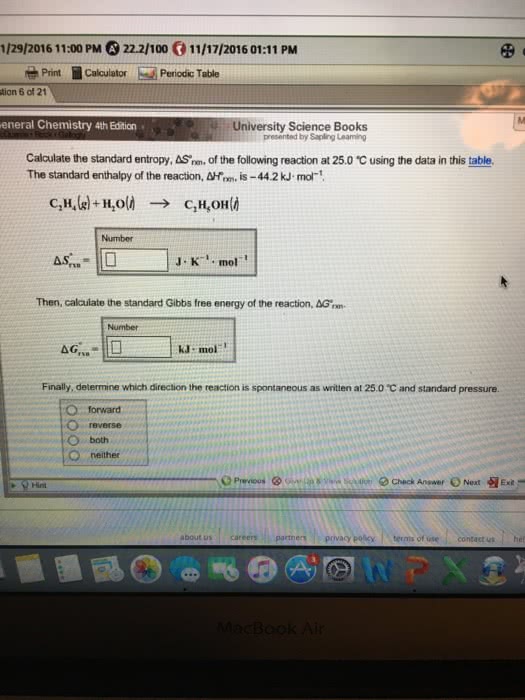
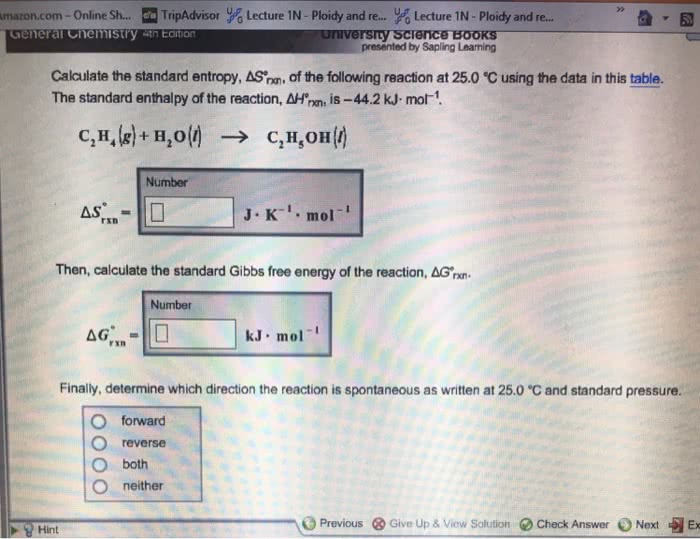
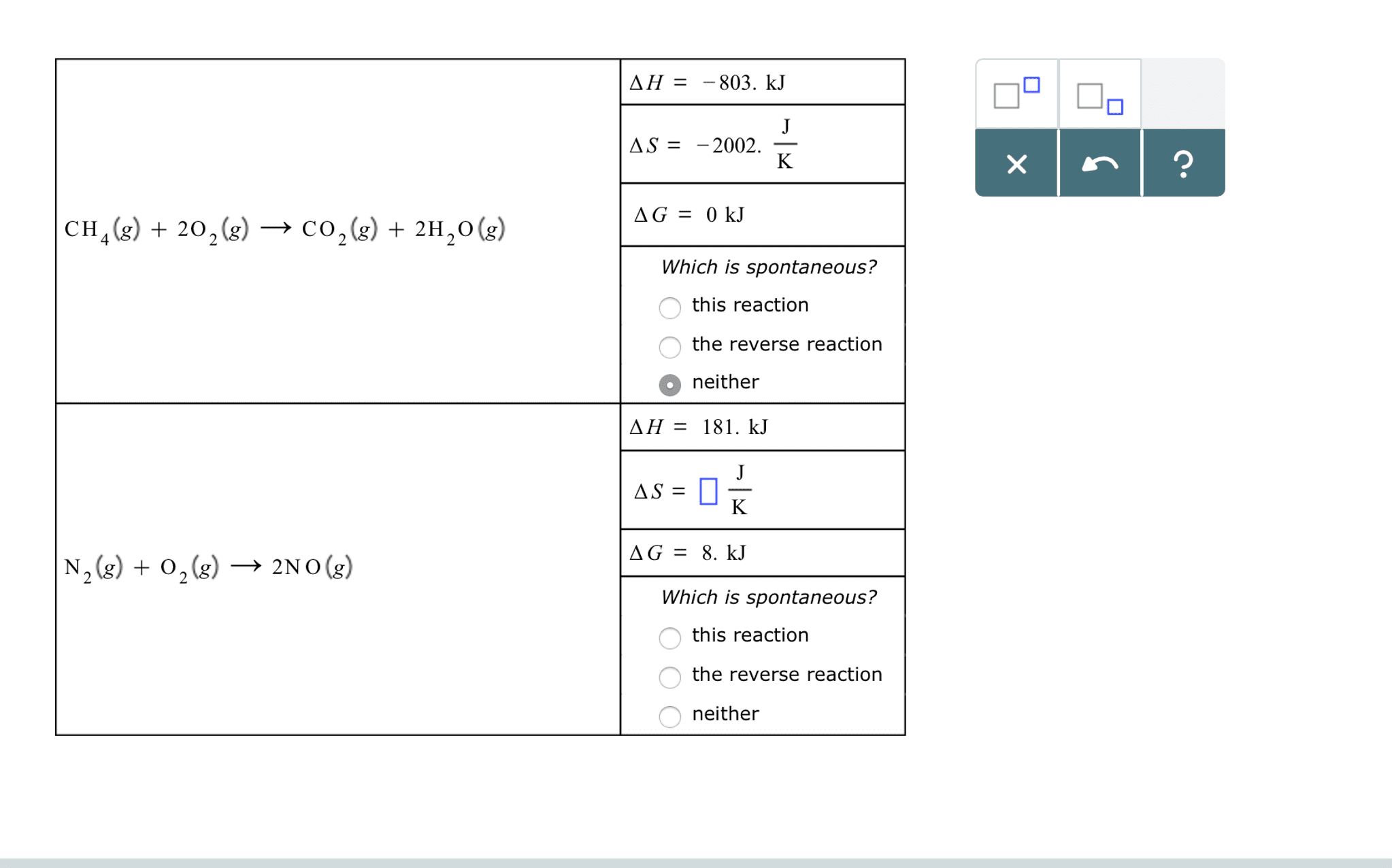
Law of conservation of energy (first law of thermodynamics) Delta g = delta h - temp x delta s, h = enthalpy, s = entropy, (-delta g = spontaneous & exergonic and + delta g = non-spontaneous & endergonic) Heat formation within a system, if h+ = endothermic and h- = exothermic. Standard free energy change has 1 m concentration and ph of 7. Keq is the ratio at equilibrium equilibrium is defined as the point where the rate of reaction in the forward direction equals the rate of reaction in the reverse direction, q=keq, delta g = 0. Q is the ratio of products at any given set up. Delta g = delta g prime + rtlnq, where q = cd/ab activation energy (ea) energy required to produce transition state. lowered by enzymes/catalyst in chemical kinetics. bicarbonate buffer. H2co3 (carbonic acid ) = h+ + hco3- (bicarbonate) buffer system in our blood.