CM-UY 1004 Lecture Notes - Lecture 5: Unified Atomic Mass Unit, Chemical Formula, Molecular Mass
31 views1 pages
25 Sep 2017
School
Department
Course
Professor
Document Summary
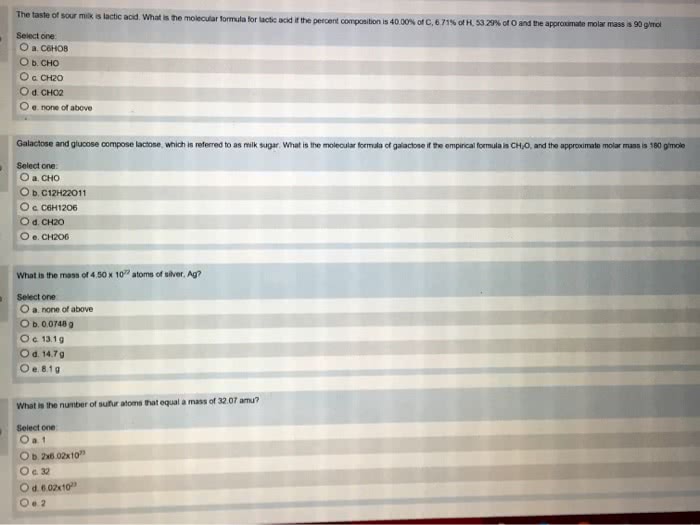
8 60 g of c h o. Molecular mass: sum of atomic masses (in amu) in a molecule. Smaller molar mass= greater number of moles. Assume 100 grams convert grams to moles by dividing my molar mass divide by smallest number of molecules empirical formula divide molar mass by mass of empirical formula for multiplier molecular formula. Chemical reaction: process where 1+ substances are changed into 1+ new substances. Chemical equation: uses chemical symbols to show what happens during a chemical.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232