CHEM 1210 Lecture Notes - Lecture 5: Pressure Measurement, Molar Volume, Combined Gas Law
Document Summary
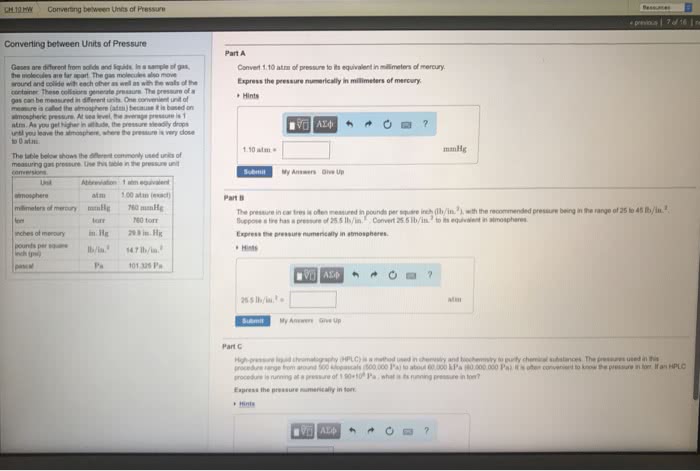
Gas pressure: pressure is force per unit area exerted against a surface (p = f/a) Most commonly measured in millimeters of mercury (mmhg), atmospheres (atm), a. Function measures atmospheric pressure measures the pressure of a confined ga. Image close together ractions olecules (atm), and torr ned gas. Boyle"s law: for a fixed mass of a gas at a constant temperature, the volume is invers proportional to the pressure. Pv = a constant or p1v1 = p2v2. Charles"s law: for a fixed volume of gas at a constant pressure, the volume is direc proportional to the temperature in kelvins (k) V/t = a constant or v1/t1 = v2/t2. Gay-lussac"s law: for a fixed mass of gas at a constant volume, the pressure is dire proportional to the temperature in kelvins (k) P/t = a constant or p1/t1 = p2/t2. Boyle"s law, charles"s law, and gay-lussac"s law can be combines 6. 022 x 1023 form.