01:160:159 Lecture Notes - Lecture 12: Rate Equation, Chemical Kinetics, Reagent

Chapter 12 - Chemical Kinetics
12.1 Reaction Rates
A. Chemical kinetics
1. Study of the speed with which reactants are converted to products
B. Reaction Rate
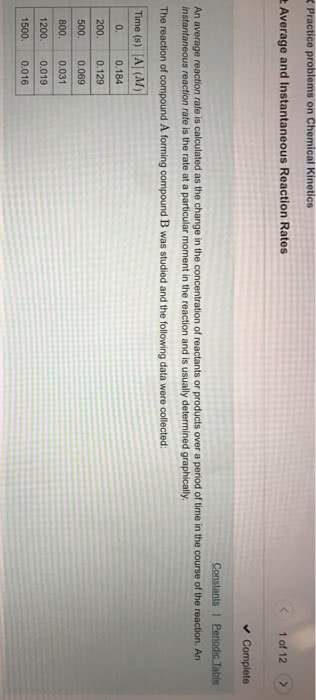
1. The change in concentration of a reactant or product per unit of time
Rate =
concentration of A at time t 2
- concentration of
A at time t1
=
D
A
t 2
- t1
Dt
a. Rates decrease with time
b. It is customary to express reaction rates as positive values
c. Instantaneous rate can be determined by finding the slope of a line
tangent to a point representing a particular
time C. Decomposition of NO2
2NO2(g) → 2NO(g) + O2(g)
Rate of consumption of NO2 = rate of production of NO = 2(rate of production of O2)
D
NO
D
æ
D
ö
2
NO
O2
-
=
=
2ç
÷
Dt
Dt
Dt
è
ø
1
find more resources at oneclass.com
find more resources at oneclass.com

12.2 Rate Laws: An Introduction
A. Reversibility of reactions
1. For the reaction 2NO2(g) → 2NO(g) + O2(g)
The reverse reaction 2NO(g) + O2(g) → 2NO2(g) may also take place
2. The reverse reaction effects the rate of change in concentrations
a. D[A] depends on the difference in the rates of the forward and
reverse reactions
B. Rate Law (Ignoring reverse reaction)
1. Rate = k[NO2]n
a. k is a proportionality constant called the rate constant
n is the order of the reactant (an integer, including zero, or a fraction)
(1) k and n must be determined experimentally
2. Concentrations of products do not appear in the rate law
C. Types of Rate Laws
1. Differential Rate Law (Rate Law)
a. Expresses how the rate of a reaction depends upon
concentration Rate = k[NO2]n
2. Integrated Rate Law
a. Expresses how the concentration of a species (reactant or product) in
a reaction depend on time
D
NO
D
æ
D
ö
2
NO
O2
-
=
=
2ç
÷
Dt
Dt
Dt
è
ø
3. Choice of rate law depends on what data is easiest to collect
***Why study rate law? Re-read the paragraph at the bottom of page 556.
12.3 Determining the Form of the Rate Law
A. Determining the value of n (Order of the reactant)
1. Example
[A]
Rate (mol/L ×s)
1.00 M
8.4 x 10-3
0.50 M
2.1 x 10-3
a. Doubling the concentration of species A quadruples the rate of the
reaction. Therefore, the reaction is of second order with respect to A
Rate = k[A]2
B. Method of Initial Rates
1. Initial rate
a. Instantaneous rate just after t = 0
2. Experimental method
a. Vary initial concentration of reactant(s)
b. Determine initial rate for each concentration
c. Examine relationship between rate and initial concentration
2
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
12. 1 reaction rates: chemical kinetics, study of the speed with which reactants are converted to products, reaction rate, the change in concentration of a reactant or product per unit of time. Dt tangent to a point representing a particular time c. decomposition of no2. Rate of consumption of no2 = rate of production of no = 2(rate of production of o2) 12. 2 rate laws: an introduction: reversibility of reactions, for the reaction 2no2(g) 2no(g) + o2(g) 2 : choice of rate law depends on what data is easiest to collect. Re-read the paragraph at the bottom of page 556. 12. 3 determining the form of the rate law: determining the value of n (order of the reactant, example. 2. 1 x 10-3: doubling the concentration of species a quadruples the rate of the reaction. Therefore, the reaction is of second order with respect to a: method of initial rates, initial rate, instantaneous rate just after t = 0, experimental method.