CHE 131 Lecture Notes - Lecture 8: Chemical Equation, Stoichiometry, Reagent
CHE 131 verified notes
8/46View all
Document Summary
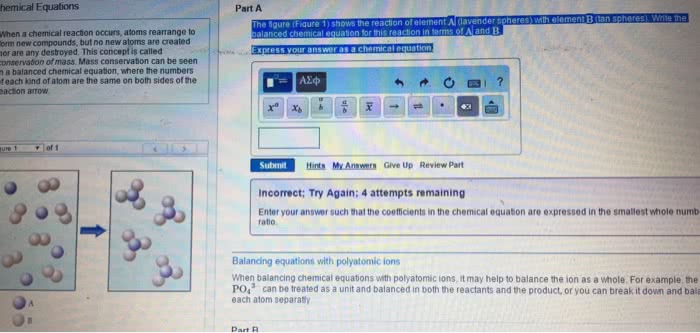
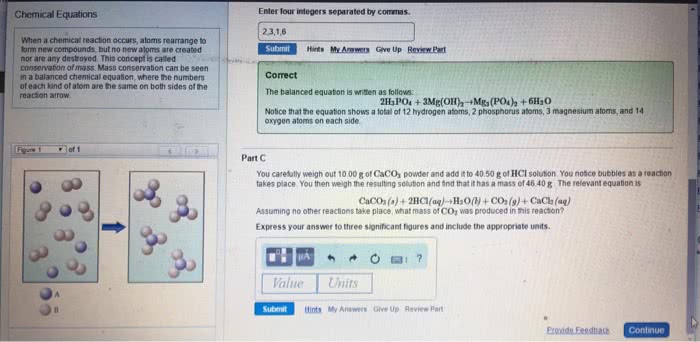
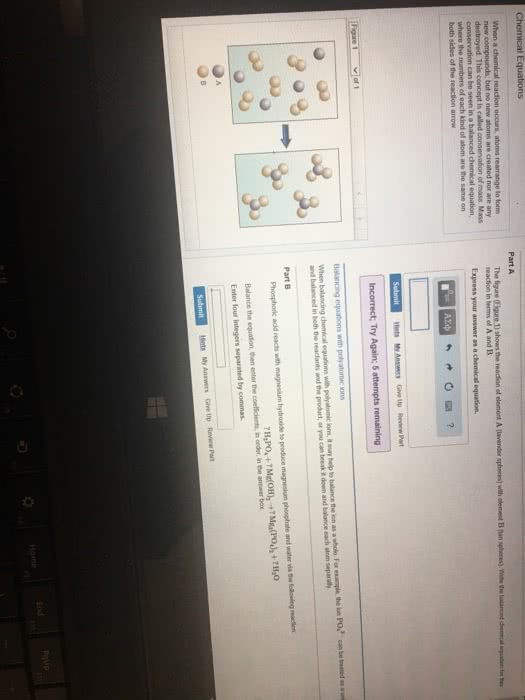
Che 131- lecture 8- chemical reactions and stoichiometry. That is, if the reactants side has 10 atoms of carbon then the products side must also have 10 atoms of carbon: that goes for total charge on both sides as well. Meaning, if you have 2so4 on the reactants side and only 1 so4 on the products side, then treat so4 as a bundle instead of thinking of the s and the o4 separately. In order to get the correct molar ratio, you need to balance the equation. This is why balancing becomes important because you can use the molar ratio for a vast majority of problems. Combustion analysis: the percent of carbon and hydrogen in hydrocarbons can be determined from the mass of h2o and co2 produced. Limiting reactant: limiting reactant- the reactant in a chemical equation that limits the amount of product that can be produced.