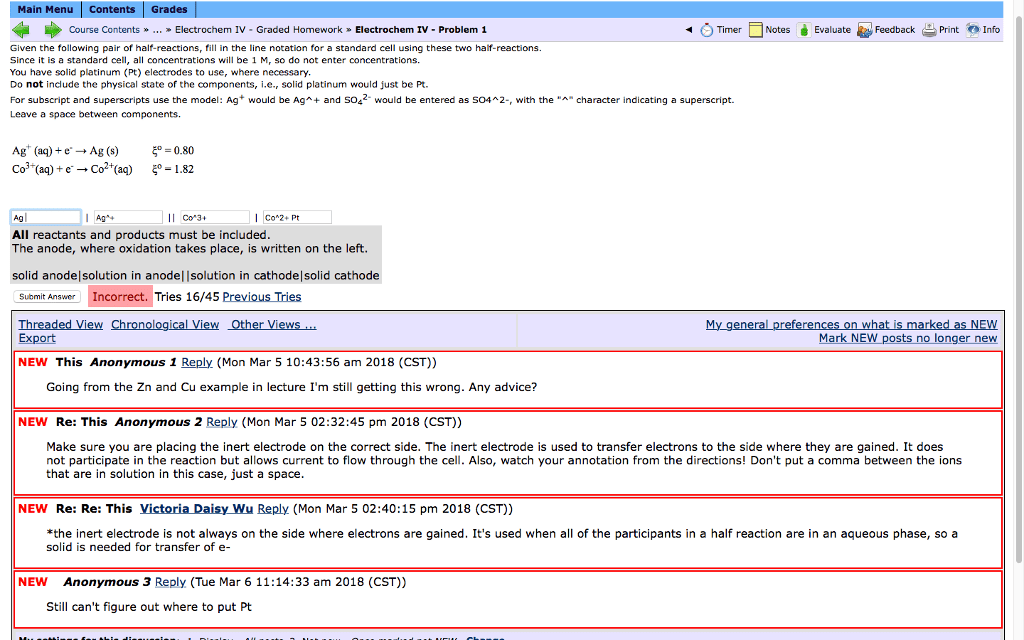
Main Menu Contents Grades Course Contents» Electrochem IV - Graded Homework> Electrochem IV-Problem 1 O Timer Notes è Evaluate-Feedback-Print e)Info Given the following pair of half-reactions, fill in the line notation for a standard cell using these two half-reactions. Since it is a standard cell, all concentrations will be 1 M, so do not enter concentrations. You have solid platinum (Pt) electrodes to use, where necessary. Do not include the physical state of the components, .e., solid platinum would just be Pt For subscript and superscripts use the model: Ag would be Ag and SO42 would be entered as S042-, with the "character indicating a superscript. Leave a space between components. Ag+ (aq) + e. â Ag (s) C03-(aq) + e. â Co2+(aq) 5°=0.80 5°-1.82 Ag Con2+ Pt Ag All reactants and products must be included The anode, where oxidation takes place, is written on the left solid anode solution in anodel Isolution in cathodelsolid cathode Submit Answer Incorrect. Tries 16/45 Previous Tries Export NE NEW This Anonymous 1 Reply (Mon Mar 5 10:43:56 am 2018 (CST)) Going from the Zn and Cu example in lecture I'm still getting this wrong. Any advice? NEW Re: This Anonymous 2 Reply (Mon Mar 5 02:32:45 pm 2018 (CST)) Make sure you are placing the inert electrode on the correct side. The inert electrode is used to transfer electrons to the side where they are gained. It does not participate in the reaction but allows current to flow through the cell. Also, watch your annotation from the directions! Don't put a comma between the ions that are in solution in this case, just a space NEW Re: Re: This Victoria Daisy Wu Reply (Mon Mar 5 02:40:15 pm 2018 (CST)) *the inert electrode is not always on the side where electrons are gained. It's used when all of the participants in a half reaction are in an aqueous phase, so a solid is needed for transfer of e- NEW Anonymous 3 Reply (Tue Mar 6 11:14:33 am 2018 (CST)) Still can't figure out where to put Pt





