BICH 410 Lecture Notes - Lecture 4: Titration Curve, Conjugate Acid, Titration
Document Summary
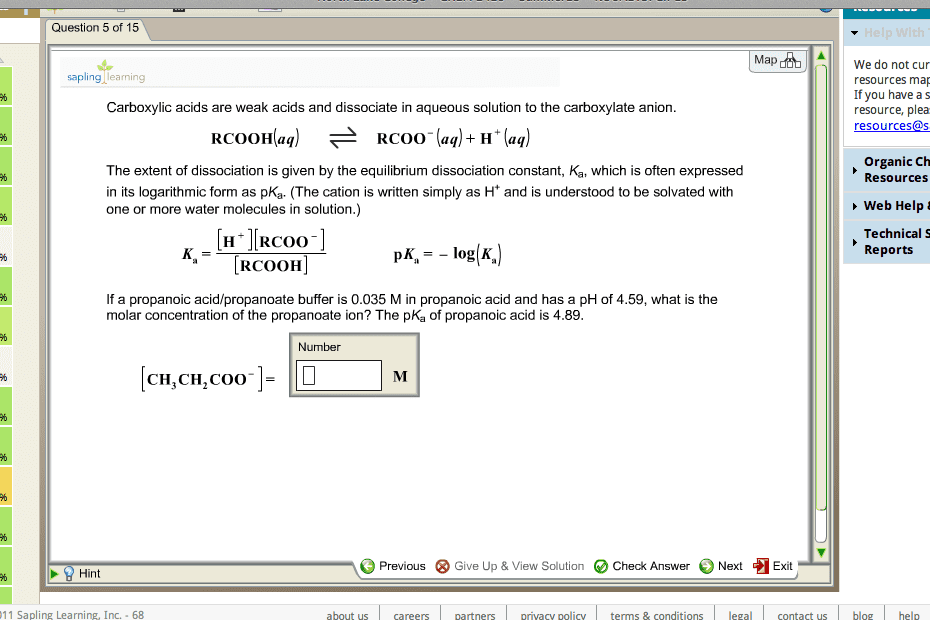
Monday, september 5, 2016 at 12:26 pm ph + poh = 14* Solutions with a ph value greater than 7 are alkaline (basic) *ph scale is logarithmic (no units) ph affects structure and function of biological macromolecules. Acid is a proton donor base is a proton acceptor. Conjugate acid-base pairs are related by the reversible reaction of a proton transfer. The strength of an acid is based on its dissociation constant or its ability to transfer a proton to water. Ka = keq pka = -log ka ph is determined by. The concentration of the conjugate acid/base pair ( how many protons are available to be donated ph = pka +log ([a-]/[ha]) henderson hasselbalch * Why is ionization of water not important for weak acids - the change amount of water is negligible. Titration curves give an experimental way to determine pka. A titration measures the amount of strong base added to an acidic solution.