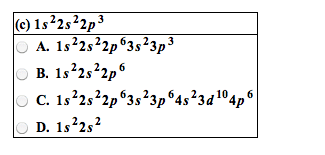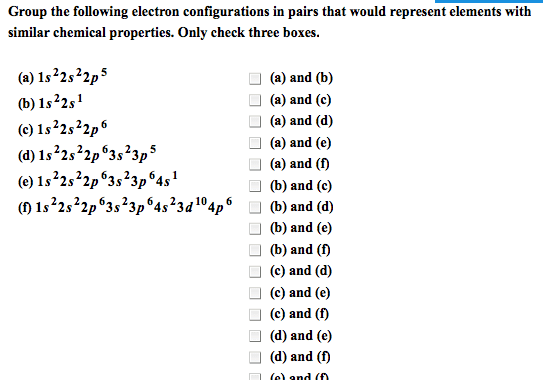CHEM 101A Lecture Notes - Lecture 3: Sodium Chloride, Fluorine, Electronegativity
Document Summary
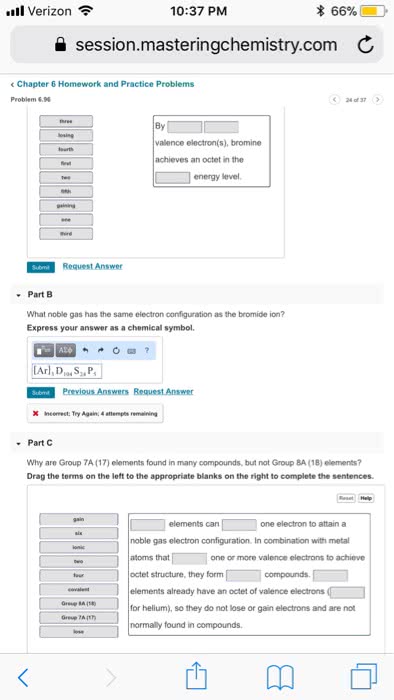
Joining of two atoms in a stable arrangement. Oxygen has two atoms- two atoms are bonded together. Always forms compounds more stable than atoms it"s made of. Only noble gases are stable as atoms. Noble gases do not readily react to form bonds. Elements gain, share, or lose electrons to get the electron configuration of a noble gas closest to it on the table. Involves only valence electron of an atom. Result from transfer of electrons from one element to another. Form between metal on left side of table and non metal on the right side. Ions: charged species where number of protons and electrons are not equal. Result from sharing of electrons between two atoms. Formed when two nonmetals combine or when metalloid bonds to a nonmetal. Molecule: compound or element containing two or more atoms joined together with covalent bonds. Consist of oppositely charged ions that have a strong electrostatic attraction- electrical attraction between oppositely charged ions.