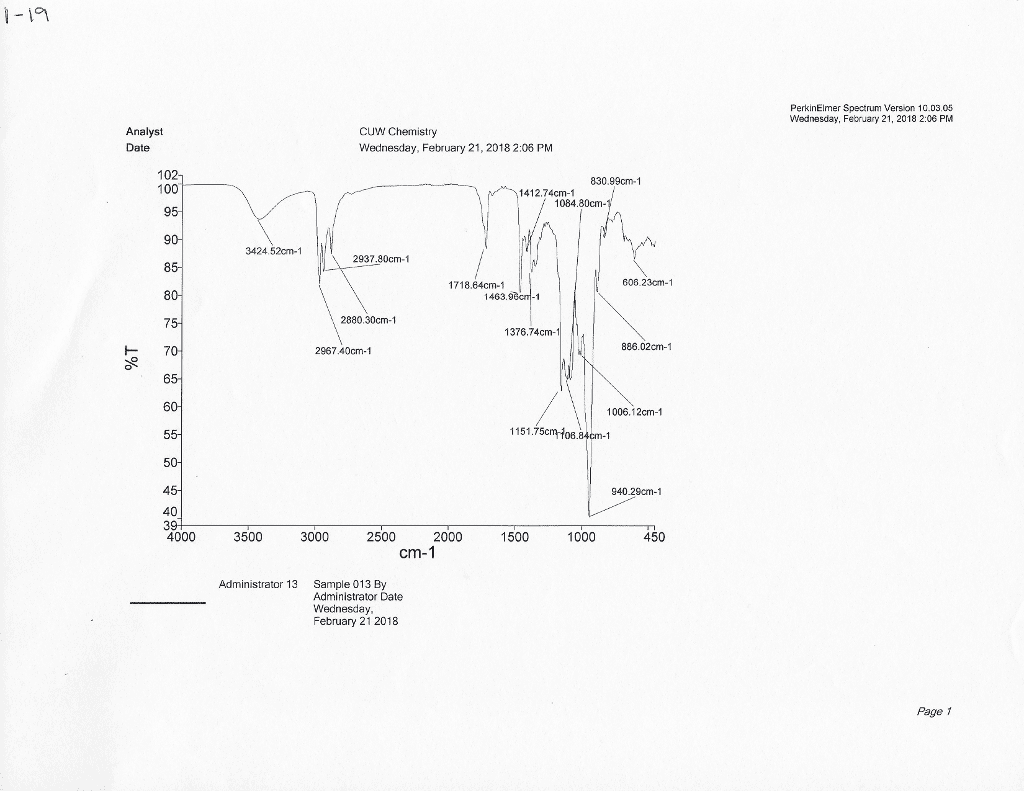
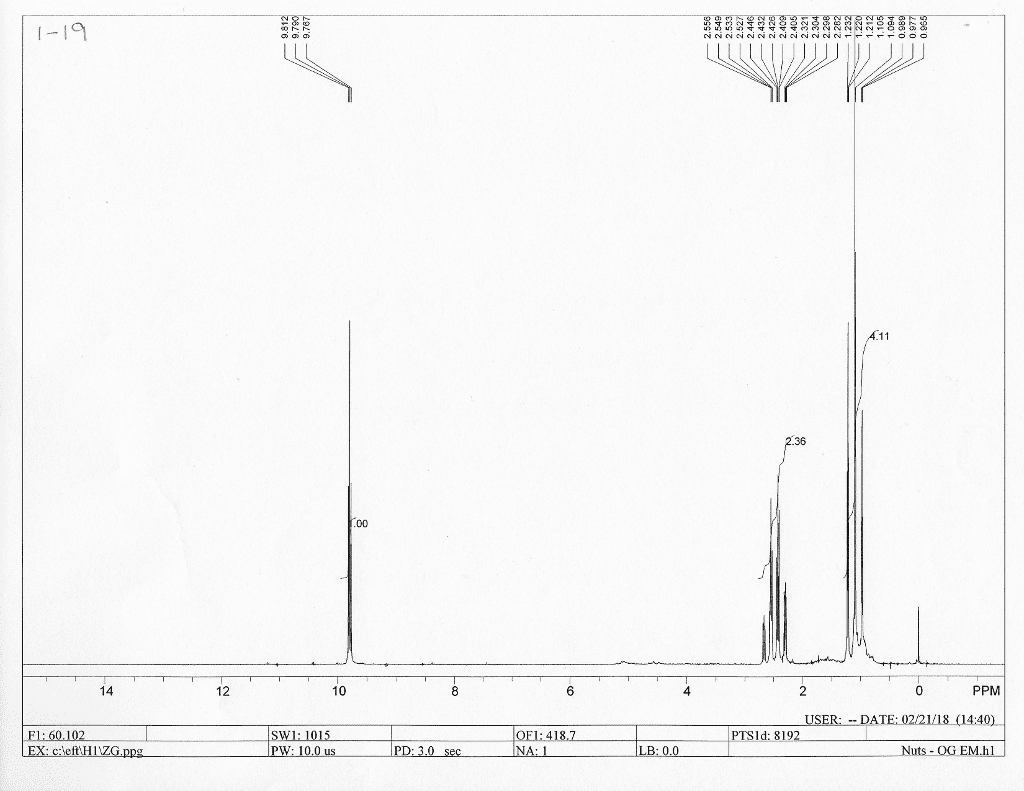
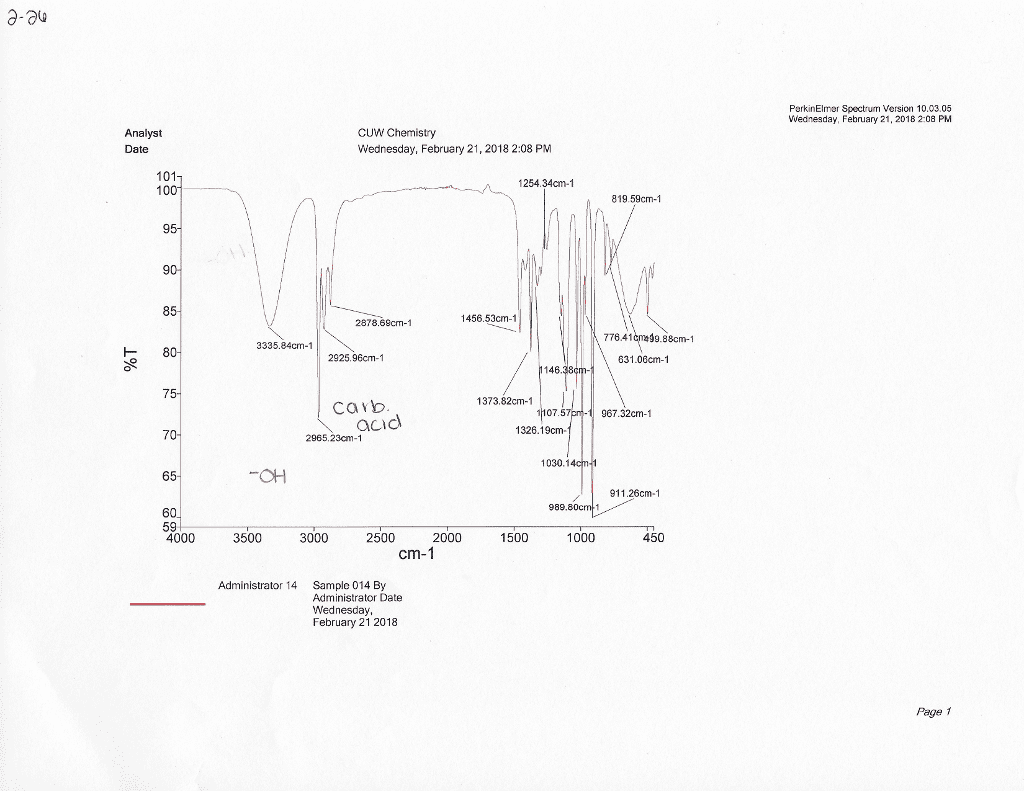
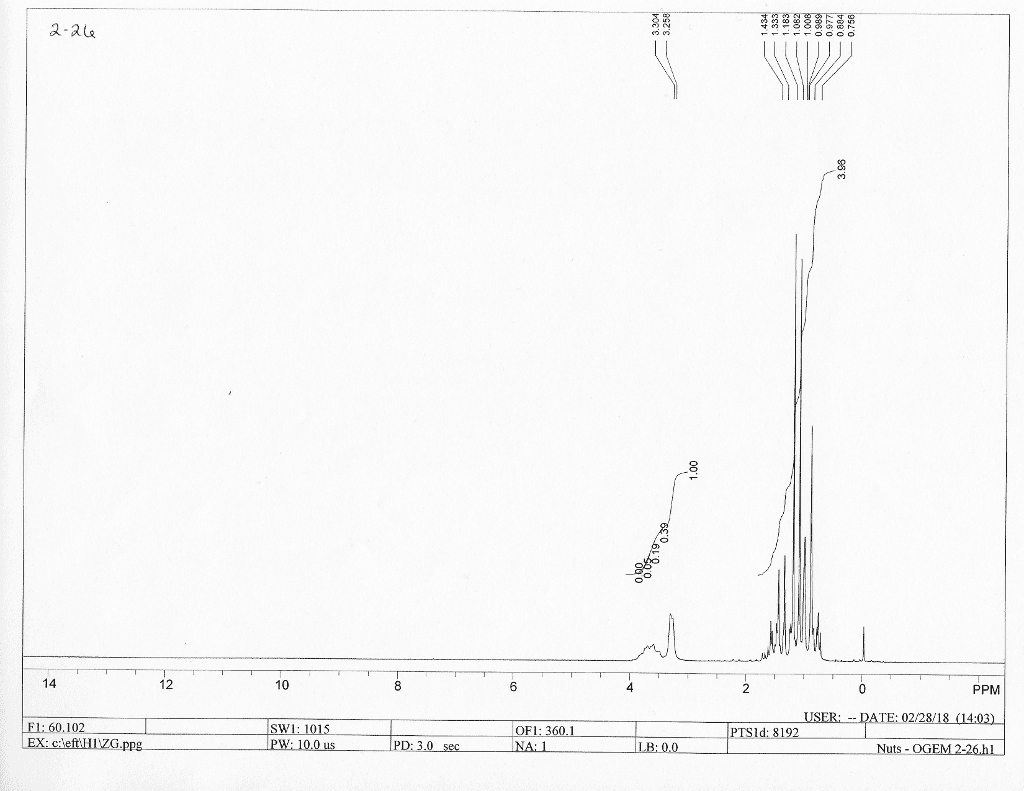
CHE 2B Lecture 2: Constant Pressure Calorimeter
Get access


Related Documents
Related Questions
Please help complete the table. Based on the experiment that was conducted. The unknown metal was placed in 40 mL of water.
| Trial 1: Time (minutes) | Trial 1: Temp (°C) | Trial 2: Time (minutes) | Trial 2: Temp (°C) | Trial 3: Time (minutes) | Trial 3: Temp (°C) | ||||
| Metal: Initial | 102 | Metal: Initial | 99 | Metal: Initial | 98 | ||||
| Calorimeter: initial | 32 | Calorimeter: initial | 34 | Calorimeter: initial | 29 | ||||
| Calorimeter + Metal: 1 min | 36 | Calorimeter + Metal: 1 min | 39 | Calorimeter + Metal: 1 min | 31 | ||||
| Calorimeter + Metal: 2 min | 42 | Calorimeter + Metal: 2 min | 40 | Calorimeter + Metal: 2 min | 36 | ||||
| Calorimeter + Metal: 3 min | 45 | Calorimeter + Metal: 3 min | 42 | Calorimeter + Metal: 3 min | 39 | ||||
| Calorimeter + Metal: 4 min | 46 | Calorimeter + Metal: 4 min | 44 | Calorimeter + Metal: 4 min | 41 | ||||
| Calorimeter + Metal: 5 min | 48 | Calorimeter + Metal: 5 min | 46 | Calorimeter + Metal: 5 min | 43 | ||||
| DT for water | DT for water | DT for water | |||||||
| DT for metal | DT for metal | DT for metal | |||||||
| Mass of water: 40 (g) | Specific Heat capacity of water (J/g°C): | ||||||||
| Calculation for Qwater: | |||||||||
| Mass of metal (g): 19 | Qmetal = - Qwater = | ||||||||
| Calculation for Csp of metal (Trial 2): | Calculation for Csp of metal (Trial 3): | |||||||
| Average Csp of metal: | Identity of Metal (extra credit): *include evidence/explanation below table | ||||||||