CHM 2041 Lecture Notes - Lecture 13: Effective Nuclear Charge, Pauli Exclusion Principle, Periodic Trends
Document Summary
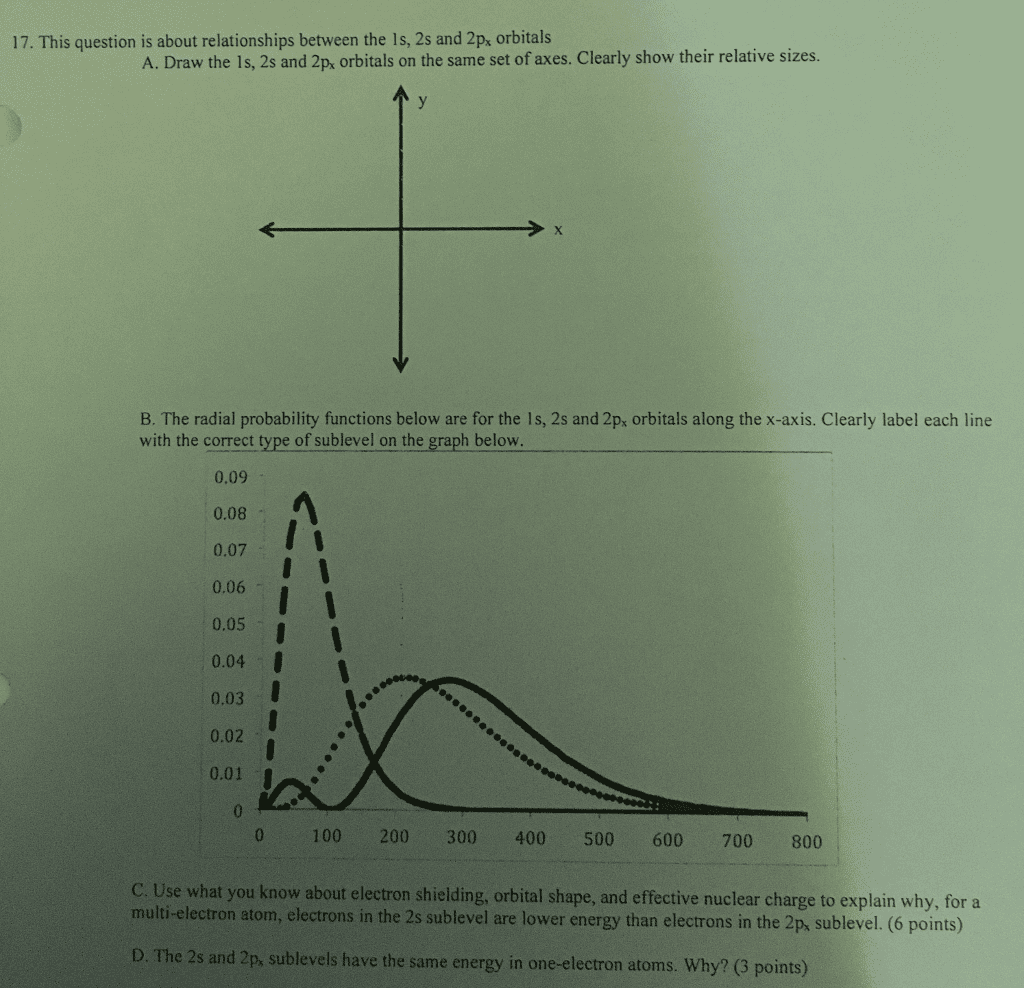
Periodic law- the elements are arranged in order of increasing mass. Electron configuration is the description of the orbitals occupies by electrons. First one is orbital and the exponent one is number of electrons in that orbital. No two electrons in an atom can have the same four quantum numbers (pauli) Energies of sublevels are split in multi-electron atoms. We call orbitals with the same energy- degenerate. Coulomb"s law (attraction and repulsion between charged particles) For like charges, potential energy (e) is positive and decreases as the. For opposite charges, the potential energy is negative and becomes. The strength of the interaction increases as size of charge increases particles get farther apart (as r increases) more negative as particles become closer which is determined by more positive nucleus. Electrons experience both repulsion of electrons and attraction of nucleus. Closer electron is to nucleus, the more attraction you will have.