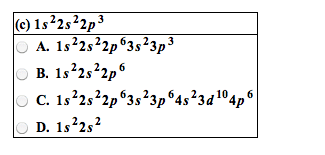CHEM 1127Q Lecture Notes - Lecture 11: Lewis Structure, Valence Electron, Transition Metal

Electronic Structure and Periodic Properties
Ions with noble gas structure
Transition Metal cations
● Transition metal cations like Ag+ and Fe3+ are different from most ions
○ Transition metal cations remove electrons from the S orbital before they remove
electrons from the D orbital.
Drawing Lewis Structures
How many electrons does an atom need?
● Lewis structures of atoms commonly forming covalent bonds
Group:
1
2
13
14
15
16
17
18
# of
valence
e-:
1
2
3
4
5
6
7
8
H
Be
B
C
N
O
F
Si
P
S
Cl
Ge
As
Se
Br
Kr
Sb
Te
I
Xe
Valence Electrons
● The number of electrons present in the outermost shell.
● Most important electrons when considering bonding.
Examples of Lewis Structures
● OH-, H2O, NH3, NH4+, C2H4

44
CHEM 1127Q Full Course Notes
Verified Note
44 documents
Document Summary
Transition metal cations like ag+ and fe3+ are different from most ions. Transition metal cations remove electrons from the s orbital before they remove electrons from the d orbital. Lewis structures of atoms commonly forming covalent bonds. The number of electrons present in the outermost shell. In certain cases, lewis structures do not represent chemical or physical reality. Both s-o bonds are equal in length, yet the lewis structure indicates one double and one single bond. Notes on resonance structures: resonance form are not different molecules, nor are they representations of electron shifting, resonance structures arise when two lewis structures are equally plausible, only electrons can be shifted in resonance structures. Formal charges are analogous to oxidation numbers: How do we know the arrangement of atoms. Cf = ve - unshared electrons - bonding electrons. Carbon = 4 - 0 - (8) = 0. Oxygen = 6 - 4 - (4) = 0.