BCH 4024 Lecture 2: L2
Document Summary
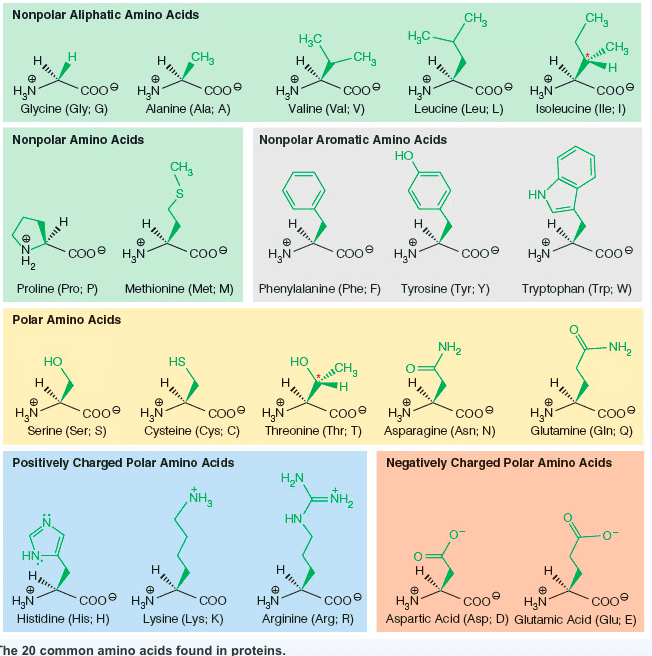
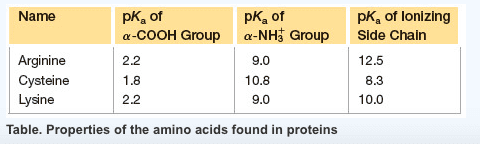
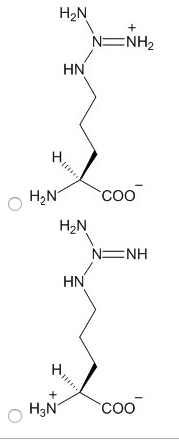
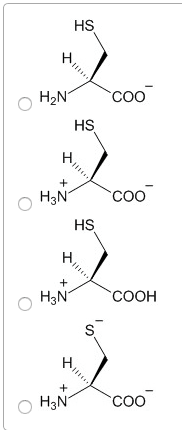
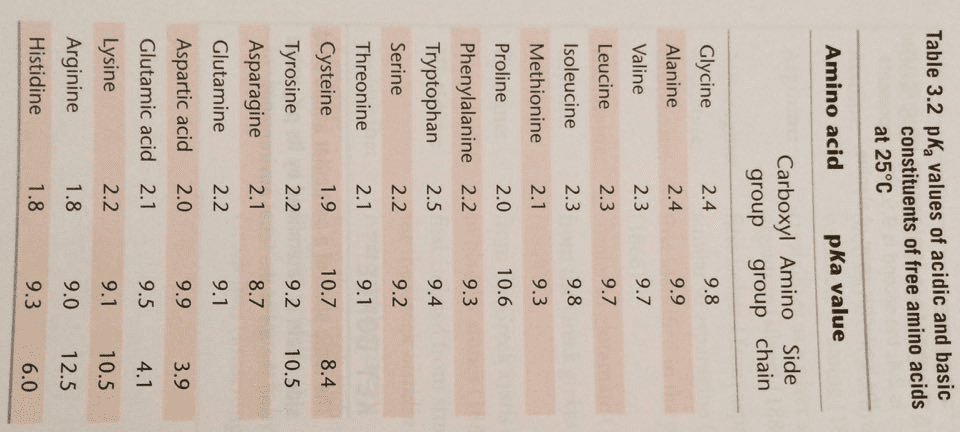
Always contain an amino group that is on alpha carbon. Greek alphabet is used to signify the alpha carbon and all side chain carbons. Stereoisomerism is based on the fischer projection for l-glyceradehyde. Tend to cluster together on the inside of proteins. All ionic forms are those at ph 7. Hydroxyl group of tyr can form hydrogen bonds and plays a catalytic role in some enzymes. Uv light absorption by aromatic amino acids and proteins. For most proteins, 1-2% of aas are aromatic. Allows us to detect the presence of aromatic molecules and to qualify their concentration. Asn & gln are amides of aspartate and glutamate. Free sh group in cysteine ionizes and is largely undissociated at physiologic ph. Sh groups oxidize to form disulfide bond in cystine. In most cases, the cytoplasm is reduced, whereas extracellular regions are oxidized. Insulin structure is stabilized by disulfide bonds. Antibody (ab): 170-kda oligomer held together by intra-chain & inter-chain disulfides.