CHM 2046 Lecture Notes - Lecture 3: Activated Complex, Collision Theory, Activation Energy
Document Summary
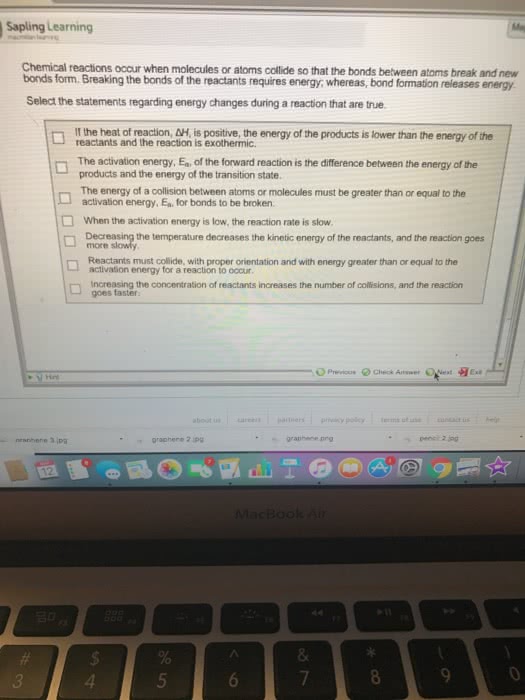
According to collision theory, for a reaction to occur molecules need to: collide with one another, collide at the correct orientation. Make sure the molecules that transfer line up: collide with enough energy to react. Increased energy can come from using more force or raising the temperature. Activation energy (ea) is the minimum amount of energy needed for a reaction to take place. Example) a + bc abc (activated complex) ab + c. The activation energy is measured from the level of reactants to the activated complex: the reverse activation energy is measured from the level of the products to the activated complex. When looking at the catalyzed reaction, the mechanism of reaction is changed from 1 to 4 because there are now four humps. Catalyzed reactions just have a different way of getting to the end of a reaction. With catalysts, more reactant is turned into product. Formula is: hrxn = hf(p) - hf(r)