CHM 2046 Lecture 1: TopHat Questions - week 1
Document Summary
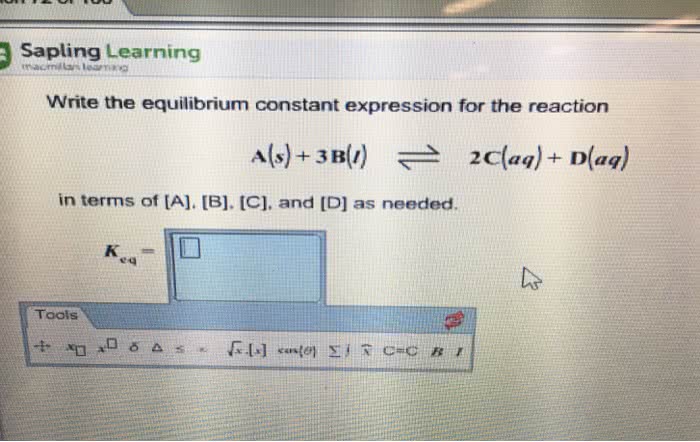
Write an equilibrium expression for a(g) + b(l) 2c(aq) + 3d(s) + e(l) 163. 9 reaction will move to reactants to reactants. 0. 25 you just had to inverse and square it to reactants. Kp was given, but the others are in mol so we need to convert kp to kc or use pv=nrt to get pressure of the equation. New kc will be 2. 5 and you get 2. 78 for q and it moves to the reactant. Q>k, the reaction will proceed to the left. Standard state is 1 mol, 1 atm, 273k (1)^2/1 and k is . 15, so it goes left! only iii. K=1 means it isn"t product or reactant favored. At k=100 you couldn"t say completion (maybe to the 8 or 9 power) At 100 k=1 and at 500 k=100, so there is more product favored and y produced at 100.