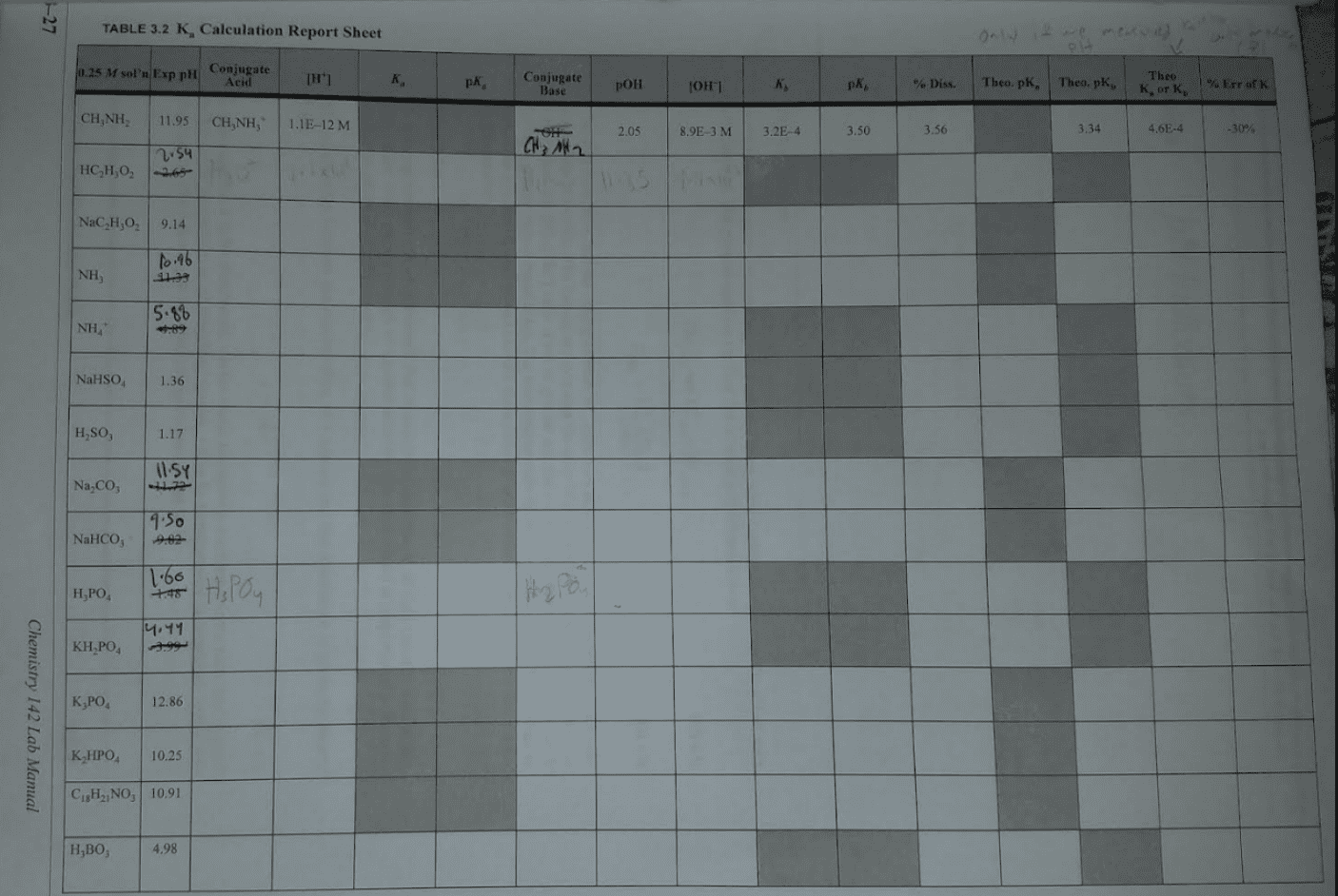
CHEM 230 Lecture Notes - Lecture 9: Acid Dissociation Constant, Equivalence Point, Conjugate Acid
82 views4 pages
Document Summary
Pug is used as a record keeping tool. One of the acid or base has to be strong. Both can be strong but both cannot be weak. We"re only concerned about the acid base reaction so we ignore na and cl. Moles of oh = 0. 01 x 0. 08 = 0. 0008. Moles of h = 0. 015 x 0. 1 = 0. 0015. We will use up of whatever has a smaller number of moles. So in this case we would use up all of the oh. To get m, divide by the total volume of 25ml to get [h+] and then use that to get ph. The equivalence point (she likes the term stoichiometric point) you added the same number of moles of acid (or base) as you have base (or acid) into your beaker. So now we"re left with 0. 0007 moles of (+ )n the beaker, we will know if it"s acid or base but we just won"t know the.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232