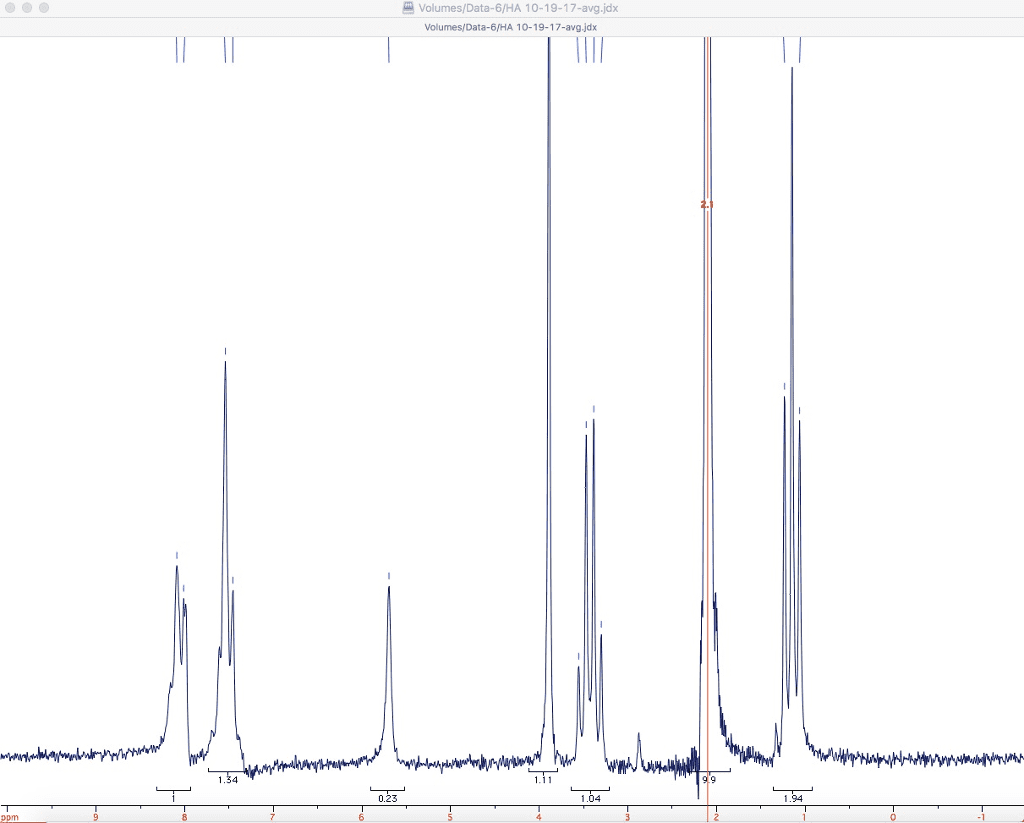
NMR (nuclear magnetic resonance) Uses the same theory and technology as an MRI!
1-What is the source of energy in NMR?____________________________________ What happens to atoms in a molecule exposed to this energy?_______________________________
2-What are two atom types that can be observed by NMR?________________________
3-What units are used to measure the strength of the magnetic field used?____________________ More significantly for us, what units are used to indicate the frequency of radiation used for resonance?________________
(Our instrument downstairs (a picoSpin) is 45 MHz. At the MN NMR Center at the U, they have instruments ranging 500 â 900 MHz.)
4-What can be distinguished with NMR?______________________________________________________
5-What are the axes of an NMR spectrum?____________________________________________________
6-What are the four different features of a spectrum that can provide information?
7-The number of NMR signals in a spectrum is equal to what?___________________________________
8-The position of an NMR signal in a spectrum depends on the electronic environment surrounding a particular __________________________.
9-If a proton is attached to an electronegative atom, it absorbs (downfield or upfield).
10-Where in an NMR spectrum would we expect to see an aldehyde H? See Table 14.1 ________________
11-What information is given by the area under an NMR signal?____________________________________
12-What information about is given by spin-spin splitting?________________________________________
13-How many peaks are contained in an NMR signal that has been described as a triplet?_________________________ How many adjacent protons are splitting this signal?________________
Skipping to section 14.11â¦
14-What do the number of different signals in a 13C spectrum indicate? __________________________________
15-What is the range of chemical shifts in a 13C spectrum?______________________________________________