CHY 183 Lecture Notes - Lecture 13: Emission Spectrum, Spectroscopy, Transition Metal
Document Summary
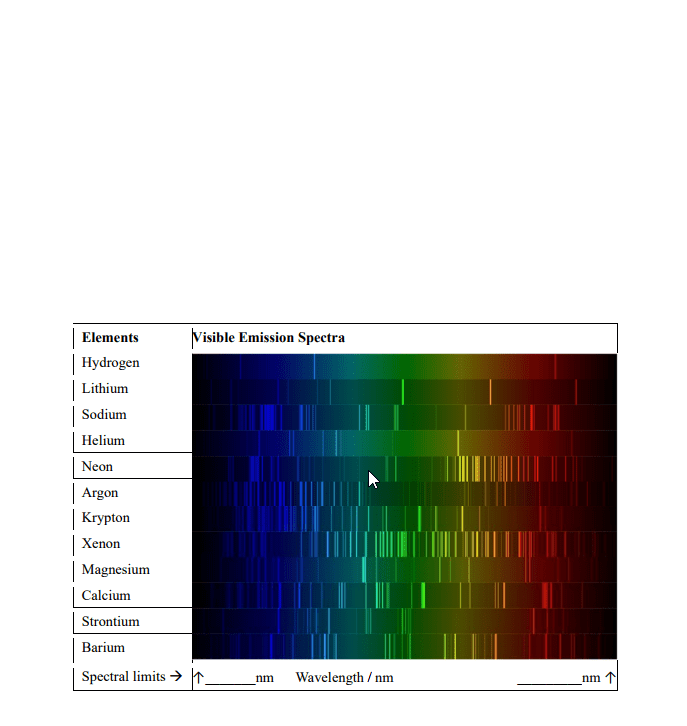
Trace elements: small quantities/concentrations of an element (<1%) Provide "invisible" markers that can establish source of material or provide additional points for comparison. Emission spectrograph: vaporizes samples by heating to a high temperature -> atoms achieve excited state -> excited atoms emit light. Emission spectrum: light emitted from a source and separated into its component colours/frequencies. Line spectrum: type of emission spectrum; shows series of lines separated by black areas; light separated into components. Can be matched line for line in a comparison between samples. Atom: nucleus (protons and neutrons) + electrons outside in electron orbitals. Orbitals associated energy level (definite amount of energy) Each elements = own set of characteristic energy levels varying in distances from the nucleus. B/c energy levels fixed, atom will absorb only a definite value of energy (heat/light) Absorbed energy pushes electrons into higher energy level orbitals; atom is now in excited state.