CHEM 123 Chapter Notes - Chapter 8: Electronegativity, Fluorine, Acid Dissociation Constant

48
CHEM 123 Full Course Notes
Verified Note
48 documents
Document Summary
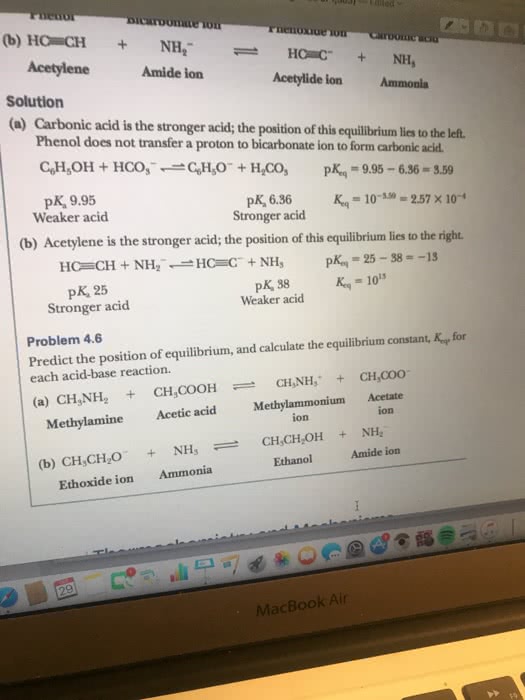
1 h - i - h conj . Whichever species l reactants / products) that"s more stable is favoured at equilibrium stronger ( acids/ bases) are less stable ( higher energy ) weaker 1 acids / bases ) are more stable ( lower energy ) Compare relative stability of } charged species. B- + ha = bh + a- need to consider when determining stability. Oxygen second most electronegative great if able to put negative charge on them. Negative charges more stable on atoms w/ higher electronegativity. Ex . than n: moreeng tess b moves towards a stable. Tiemeyer l for same type of atom ) Electronegativity useful when comparing ions / atoms bearing charges in same row of pt. Key concepts: the more en- the atom , the more it can stabilize , the more en- the atom , the more destabilized by lie on side of more stable species, equilibrium will.