CHEM261 Lecture 4: CHEM261 Lecture : Sept 29 2022 Chem 261 notes

1
CHEM261 Full Course Notes
Verified Note
1 document
Document Summary
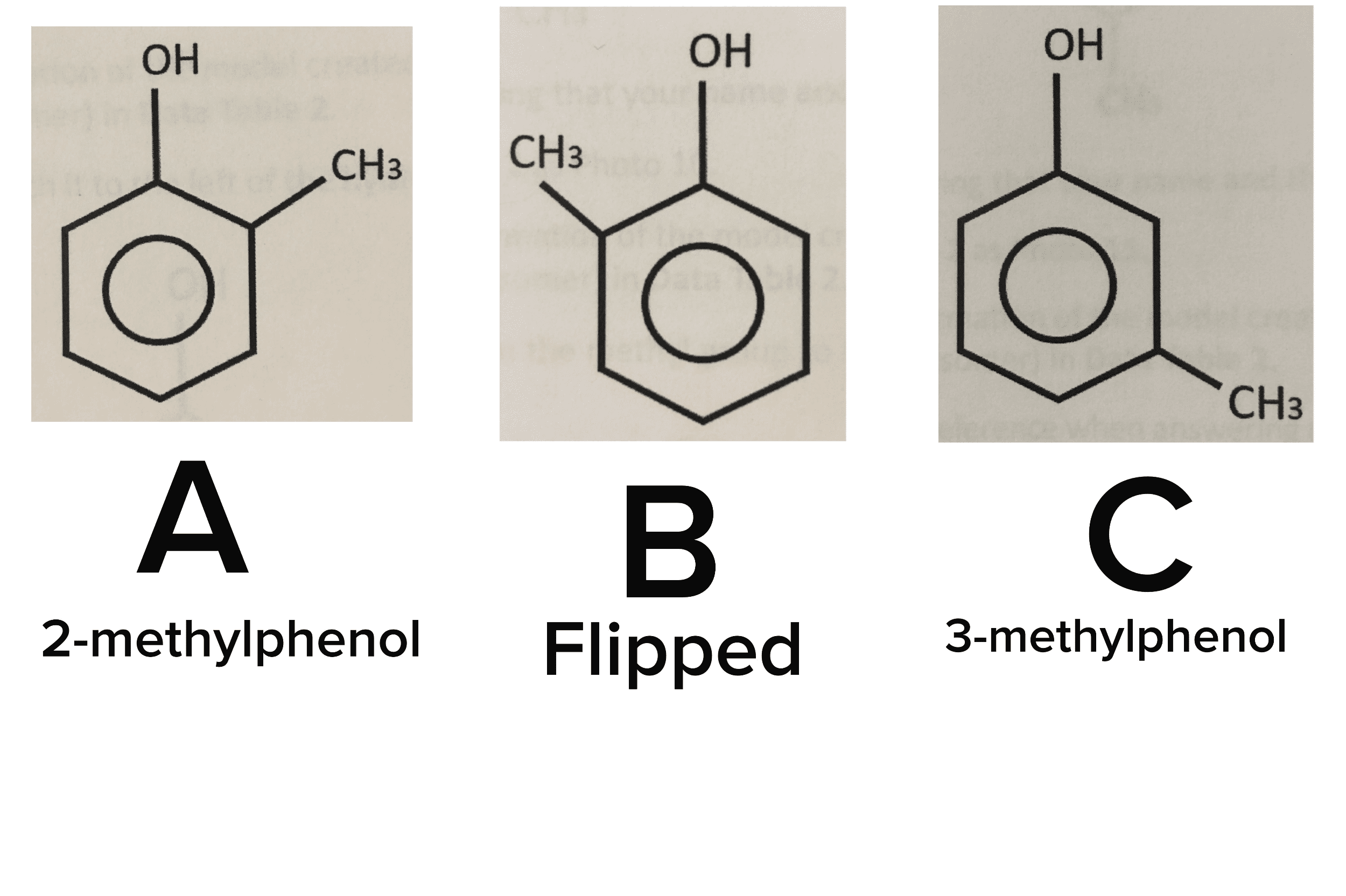
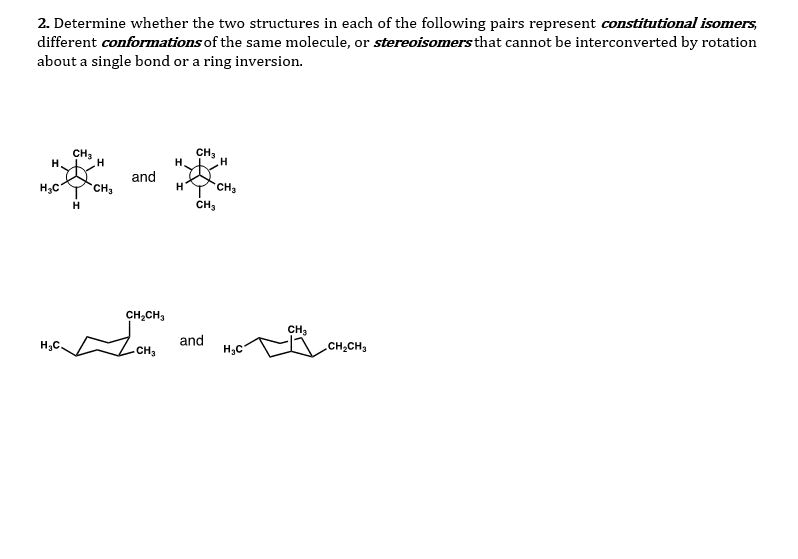
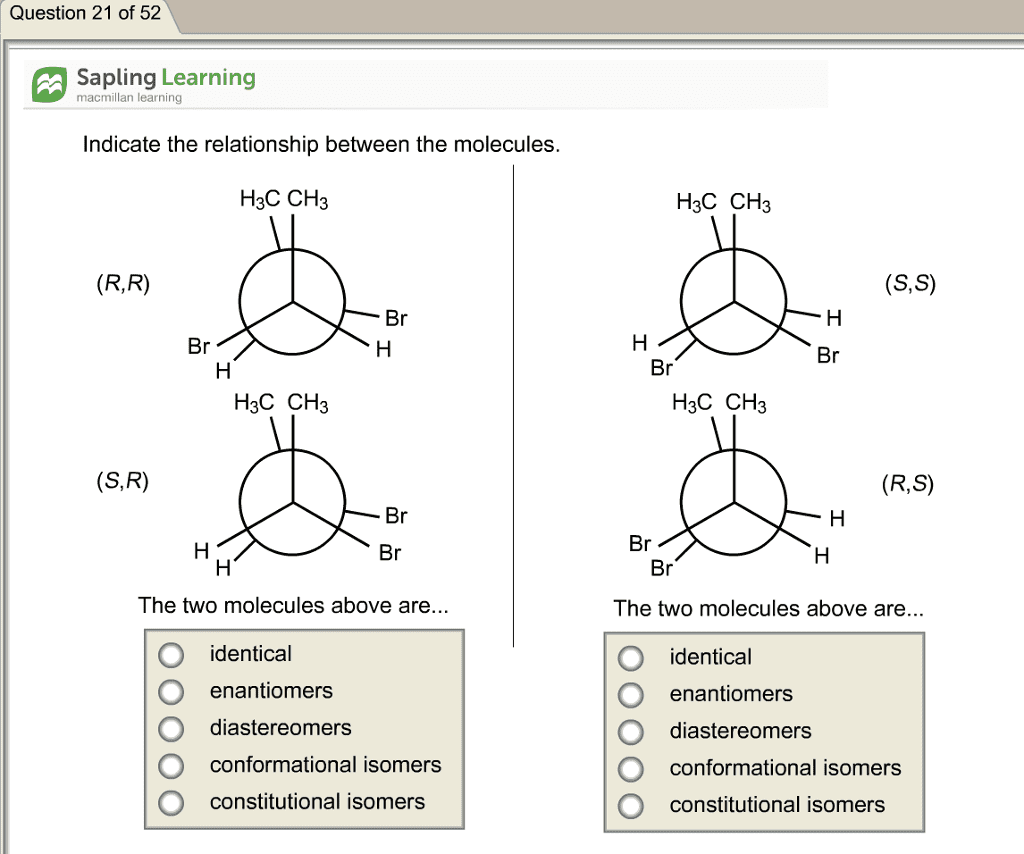
Isomers are compounds with the same molecular formula & a different arrangement of atoms in the molecule. Different 3d shapes of a single (the same) molecule obtained by rotation about single bonds. A molecular conformation is any spatial arrangement of atoms that can be interconverted by rotation about single bonds of the molecule. At room room temperature (20 oc): 15-20 kcal/mol of energy available. This allows for rotation around c-c to occur rapidly at room temperature. There is a steric effect between neighboring bonds to hydrogens: repulsion of filled shells of e- This is a tool to examine the conformation (rotational 3d geometry) about one specific bond. Look at one bond at a time. Back c is a large circle and the front c is a dot back atoms front atoms. Staggered conformation (hydrogens are anti) eclipsed conformation (hydrogens are syn) Anti means opposite side syn means same side. Needs energy to go from one conformation to another.