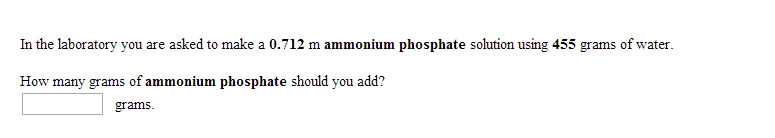1
answer
0
watching
362
views
28 Sep 2019
1.An aqueous solution is made by dissolving 24.6 grams of cobalt(II) nitrate in 342 grams of water. The molality of cobalt(II) nitrate in the solution is ___m.
2. In the laboratory you are asked to make a 0.639 m sodium sulfide solution using 425 grams of water. How many grams of sodium sulfide should you add?
___ grams
3. In the laboratory you are asked to make a 0.550 m ammonium chloride solution using 14.8 grams of ammonium chloride. How much water should you add? ___ grams
1.An aqueous solution is made by dissolving 24.6 grams of cobalt(II) nitrate in 342 grams of water. The molality of cobalt(II) nitrate in the solution is ___m.
2. In the laboratory you are asked to make a 0.639 m sodium sulfide solution using 425 grams of water. How many grams of sodium sulfide should you add?
___ grams
3. In the laboratory you are asked to make a 0.550 m ammonium chloride solution using 14.8 grams of ammonium chloride. How much water should you add? ___ grams
Reid WolffLv2
29 Sep 2019