1
answer
1
watching
4,776
views
28 Sep 2019
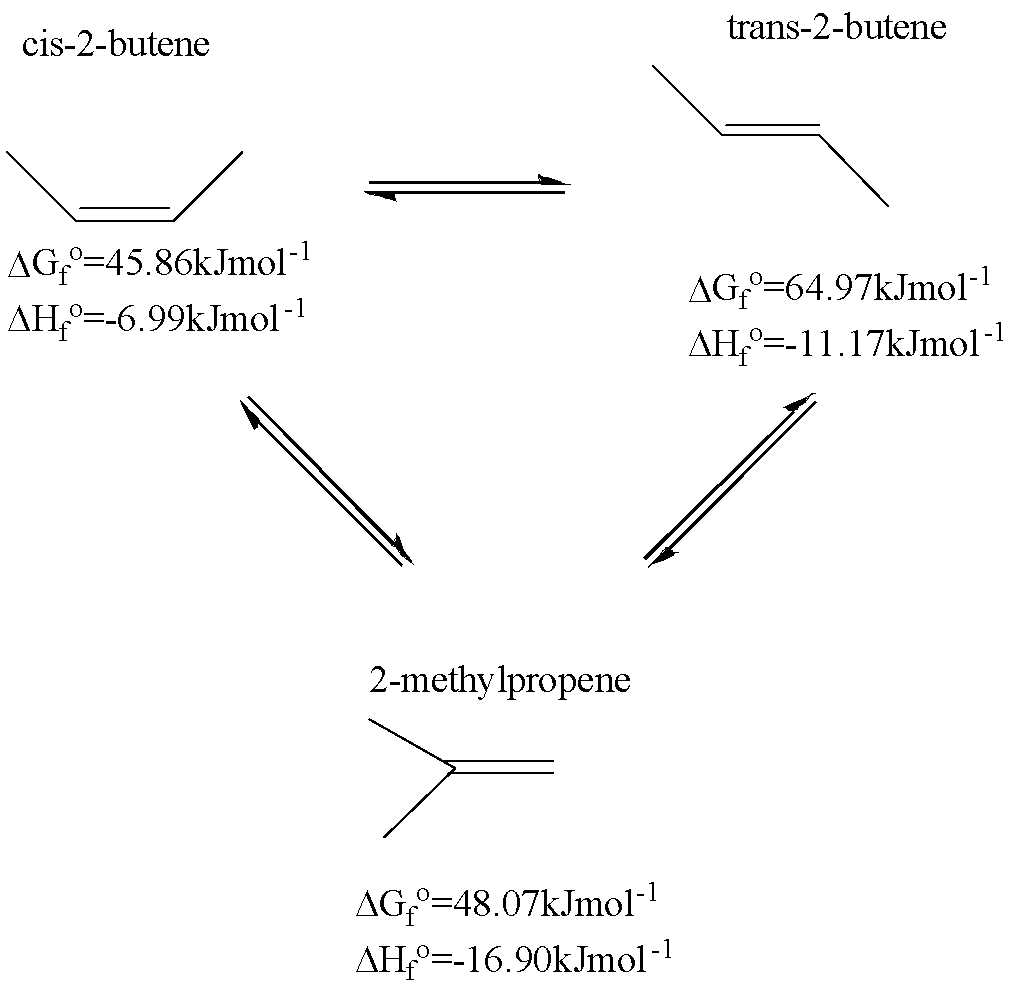
Calculate the delta H reaction for the combustion of glucose(C6H12O6) using delta H(f knot) values. Calculate the delta H(fknot) of glucose to be used in the above calculation using thefollowing information. (must be calculated, not looked up).
6CO2(g) + 6H2O(l) = C6H12O6(s) + 6O2(g) Delta H (rxn)= 2826 kJ
* Please show all work and steps so I can understand how to dosimilar probelms*
Thank you!
Calculate the delta H reaction for the combustion of glucose(C6H12O6) using delta H(f knot) values. Calculate the delta H(fknot) of glucose to be used in the above calculation using thefollowing information. (must be calculated, not looked up).
6CO2(g) + 6H2O(l) = C6H12O6(s) + 6O2(g) Delta H (rxn)= 2826 kJ
* Please show all work and steps so I can understand how to dosimilar probelms*
Thank you!
Verified Answer
Reid WolffLv2
30 Sep 2019
Joey Tang
Bachelor’s Degree in Chemistry from McMaster University19 May 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.