3
answers
0
watching
154
views
azurecrow764Lv1
28 Sep 2019
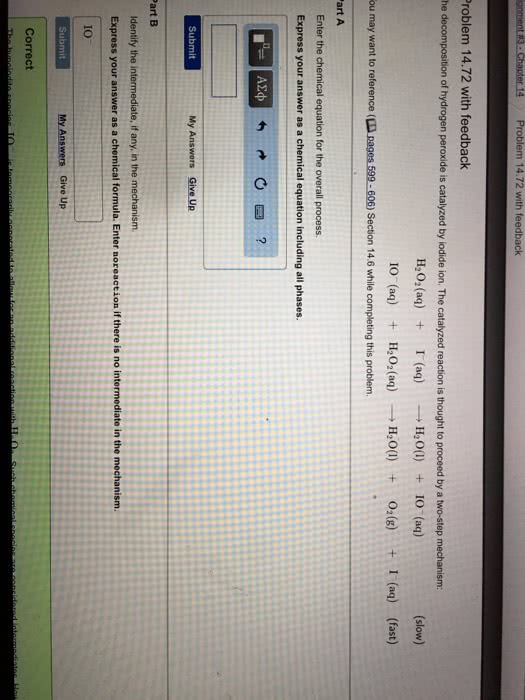
The decomposition of hydrogen peroxide can be catalyzed byiodide ion. The catalyzed reaction is thought to proceed by a twostep mechanism.
step 1 H2O2(aq) + I-(aq) => H2O(l) + IO-(aq)
step 2 IO-(aq) +H2O2(aq) => H2O(l) +I-(aq)
a. Write the rate law for each of the elementary processes ofthe mechanism.
b. Write the chemical equation for the overall reaction.
c. Identify the intermediate, if any, in the mechanism.
d. If the rate law is Rate= k [H2O2][I-] which step is slow?
The decomposition of hydrogen peroxide can be catalyzed byiodide ion. The catalyzed reaction is thought to proceed by a twostep mechanism.
step 1 H2O2(aq) + I-(aq) => H2O(l) + IO-(aq)
step 2 IO-(aq) +H2O2(aq) => H2O(l) +I-(aq)
a. Write the rate law for each of the elementary processes ofthe mechanism.
b. Write the chemical equation for the overall reaction.
c. Identify the intermediate, if any, in the mechanism.
d. If the rate law is Rate= k [H2O2][I-] which step is slow?
shitalbhusare12Lv10
24 Mar 2022
Already have an account? Log in
Hubert KochLv2
30 Sep 2019
Already have an account? Log in