1
answer
1
watching
1,365
views
5 Nov 2019
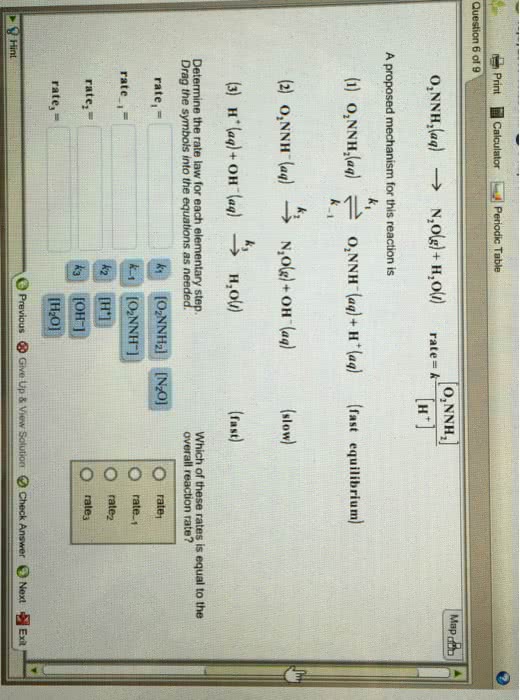
The decomposition of nitramide, O2NNH2, in water has the following chemical equation and rate law. O2NNH2 (aq) yields N2O (g) + H2O (l) rate= [O2NNH2]/[H]A proposed mechanism for this reaction is.k1(1) O2NNH2 (aq) equilibrium O2NNH (aq) + H (aq) (fast equilibrium)k-1k2(2) O2NNH (aq) yields N2O (g) + OH (aq) (slow)k3(3) H (aq) + OH yields H2O (l) (fast)What is the relationship between the observed value of k and the rate constants for the individualsteps of the mechanism?k=__________Also which symbols go into the numerator or demoniator k-1, k3, k2, k1
The decomposition of nitramide, O2NNH2, in water has the following chemical equation and rate law. O2NNH2 (aq) yields N2O (g) + H2O (l) rate= [O2NNH2]/[H]A proposed mechanism for this reaction is.k1(1) O2NNH2 (aq) equilibrium O2NNH (aq) + H (aq) (fast equilibrium)k-1k2(2) O2NNH (aq) yields N2O (g) + OH (aq) (slow)k3(3) H (aq) + OH yields H2O (l) (fast)What is the relationship between the observed value of k and the rate constants for the individualsteps of the mechanism?k=__________Also which symbols go into the numerator or demoniator k-1, k3, k2, k1
Verified Answer
Casey DurganLv2
21 Apr 2020
Joey Tang
Bachelor’s Degree in Chemistry from McMaster University24 Jun 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.