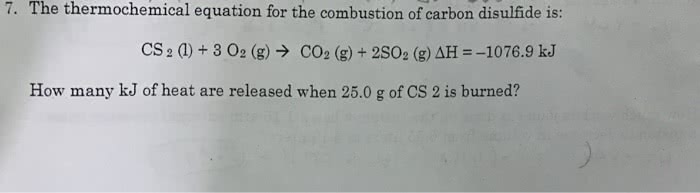2
answers
0
watching
512
views
10 Jun 2018
59. Consider the thermochemical equation for the combustion of acetone (CHO), the main ingredient in nail polish remover: C3H2O(l) + 4 O2(g) — 3 CO2(g) + 3 H2O(g) AHx= -1790 kJ If a bottle of nail polish remover contains 177 ml of acetone, how much heat is released by its complete combustion? The density of acetone is 0.788 g/mL.
59. Consider the thermochemical equation for the combustion of acetone (CHO), the main ingredient in nail polish remover: C3H2O(l) + 4 O2(g) — 3 CO2(g) + 3 H2O(g) AHx= -1790 kJ If a bottle of nail polish remover contains 177 ml of acetone, how much heat is released by its complete combustion? The density of acetone is 0.788 g/mL.
Hubert KochLv2
10 Jun 2018
Already have an account? Log in