1
answer
0
watching
4,052
views
8 Feb 2019
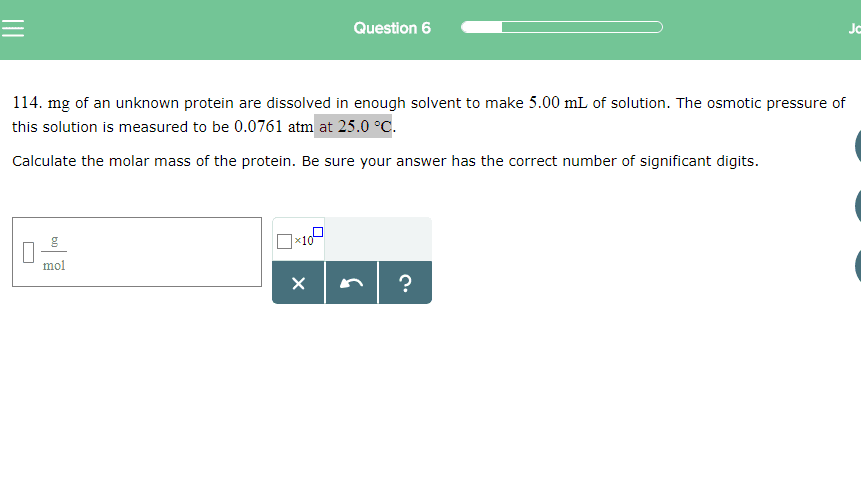
A protein subunit from an enzyme is part of a research study and needs to be characterized. A total of 0.150 g of this subunit was dissolved in enough water to produce 2.00 mL of solution. At 28 ∘C the osmotic pressure produced by the solution was 0.138 atm. What is the molar mass of the protein?
A protein subunit from an enzyme is part of a research study and needs to be characterized. A total of 0.150 g of this subunit was dissolved in enough water to produce 2.00 mL of solution. At 28 ∘C the osmotic pressure produced by the solution was 0.138 atm. What is the molar mass of the protein?
Beverley SmithLv2
9 Feb 2019