1
answer
0
watching
141
views
28 Sep 2019
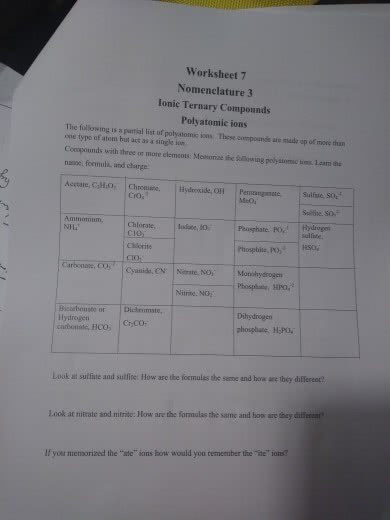
Why do some compounds with polyatomic ions have parentheses and can you show me an example?
Why do some compounds with polyatomic ions have parentheses and can you show me an example?
1
answer
0
watching
141
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Hubert KochLv2
28 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
What is the total concentration of all ions (the sum of their individual concentrations) in aqueous solutions of the following?
Hint: Each substance is an electrolyte that dissociates in water to produce ions. Use the link table of polyatomic ions if you do not recognize the polyatomic ion. For example, Na2SO4 -> 2Na1+ + SO42- when it dissolves, so a 1 M soln of Na2SO4 is 2M in Na1+ and 1M in SO42- and the total concentration of ions is 2 M + 1 M = 3M.
| 0.0760-M CaCl2 | _____________ M |
| 0.0160-M Na2SO4 | _____________ M |
| 0.0740-M K2Cr2O7 | _____________ M |